Design & Development That Keeps Every Requirement Linked, Documented, and Defensible.
Full traceability from requirements through validation — built for every regulated industry.
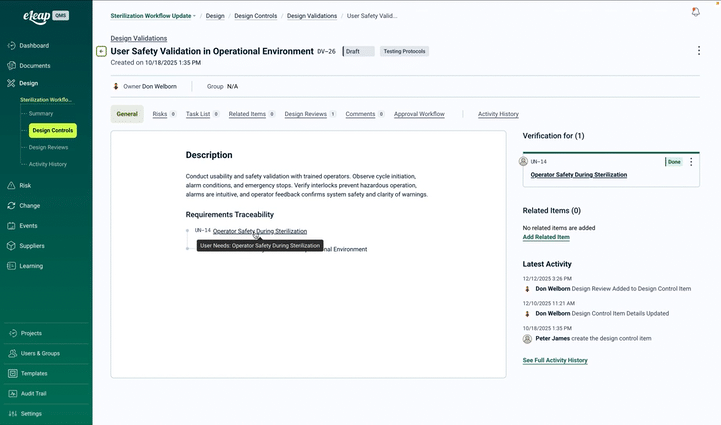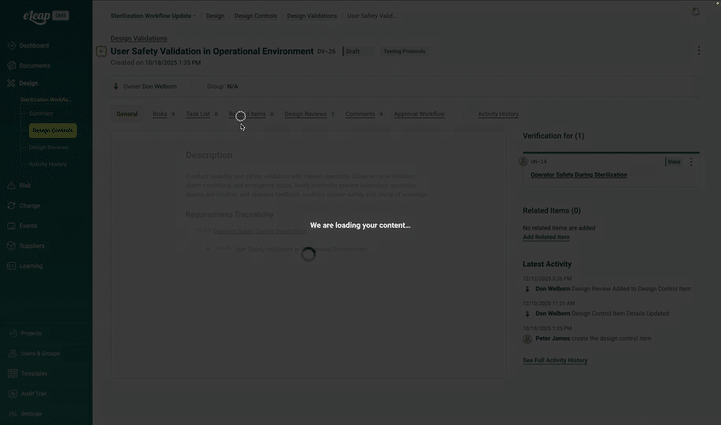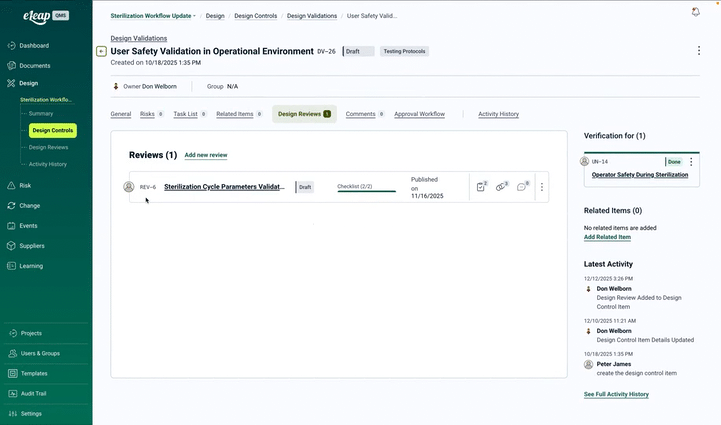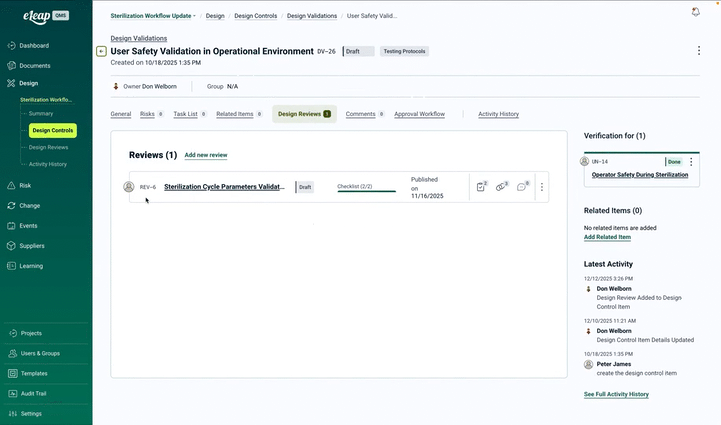

The Design & Development Problem: Book a Demo
Whether you work in medical devices, aerospace, automotive, pharmaceuticals, or any other regulated industry, the design and development challenge is the same: requirements, specifications, and test results scattered across disconnected systems — and documentation that is never quite ready when you need it.
- ❌ Technical documentation packages currently take months of manual labor to compile after development is complete — instead of generating automatically as work progresses.
- ❌ Manually maintaining bi-directional traceability across user needs, technical specifications, and test results leads to errors that trigger regulatory findings and audit observations.
- ❌ Design and development changes trigger a manual cascade of updates across disconnected spreadsheets and documents — increasing the risk of obsolete data and missed re-verifications.
- ❌ Inadequate design and development controls are a leading cause of delayed regulatory approvals, certification failures, and costly late-stage rework across all regulated industries.
- ❌ Training on updated specifications and procedures is tracked separately from the development record — creating a competency gap that regulators and auditors consistently flag.
The eLeaP Solution:
- ✅ Compile audit-ready design and development records — including history packages, technical files, and development dossiers — as you work, not months after development is complete.
- ✅ Eliminate manual traceability spreadsheets with a system that automatically identifies orphaned requirements and incomplete verifications in real time.
- ✅ Seamlessly link risk analysis to design outputs per ISO 14971, ICH Q9, AS9100, or IATF 16949 — ensuring hazards and controls are always synchronized with the development record.
- ✅ Purpose-built for regulated industries: ISO 13485, AS9100, IATF 16949, QMSR (21 CFR Part 820), ICH guidelines, ISO 9001, and cGMP requirements — in one unified platform.
- ✅ The only Design & Development system with Enterprise LMS built in — training cascades automatically as specifications change, closing the competency gap that generic QMS platforms ignore.
Stop risking regulatory delays and certification failures due to inadequate design and development controls. eLeaP’s Design & Development module provides a comprehensive framework for managing product and process development from concept through commercialization — across every regulated industry. → Talk to an Expert.
Transforming Product Development in Regulated Industries
Whether you are developing medical devices, pharmaceutical products, aerospace components, automotive parts, biologics, or regulated software, design and development controls are critical for regulatory approval, certification, and market success. Traditional approaches using disconnected spreadsheets and documents create traceability gaps that lead to audit observations, certification delays, and costly field actions.
eLeaP’s Design & Development System delivers comprehensive compliance without sacrificing development speed. At its core is a flexible module that adapts to your specific regulatory requirements and development methodology — waterfall, stage-gate, agile, or hybrid. With integrated risk management, automated verification and validation protocols, and seamless technical documentation compilation, eLeaP ensures your products and processes meet global regulatory requirements while reducing time to market.
Key Features
-
Configurable Development Framework
Adapt the system to your development methodology — waterfall, agile, stage-gate, or hybrid. Configure phase gates, deliverables, and approval requirements based on product classification, regulatory pathway, and industry standard.
-
Requirements Management System
Capture and control all requirements including user needs, regulatory standards, customer specifications, and technical requirements. Maintain bi-directional traceability with automatic impact analysis for changes across every phase.
-
Dynamic Traceability Matrix
Automatically generate and maintain traceability from requirements through design outputs, testing, and validation. Instantly identify gaps, orphaned requirements, and incomplete verifications — with no manual spreadsheet maintenance.
-
Design Review Management
Configure stage-gate reviews with role-based participants, customizable checklists, and action tracking. Support formal design reviews, technical reviews, and agile sprint reviews with complete, audit-ready documentation.
-
Verification & Validation Suite
Create, execute, and approve test protocols with electronic signatures. Link results directly to requirements and automatically flag deviations for investigation. Support all testing types from bench testing through process validation and clinical studies.
-
Risk Management Integration
Integrate risk analysis throughout development per ISO 14971, ICH Q9, AS9100, IATF 16949, or your specific framework. Track hazards, controls, and residual risk with automatic documentation updates linked to your development record.
-
Technical Documentation Automation
Automatically compile design and development history records, technical documentation packages, and regulatory dossiers based on your industry requirements — for FDA, ISO, IATF, AS9100, or other regulatory bodies.
-
Integrated Training Cascade
When specifications change, eLeaP automatically identifies affected personnel and deploys targeted training assignments — with completion tracking and competency verification built into the development record.
-
Development Transfer Protocols
Ensure successful scale-up and commercialization with transfer protocols, process validation requirements, and manufacturing readiness assessments linked directly to your development record.
Why Select eLeaP Quality?
eLeaP is the only Design & Development system with Enterprise Learning Management built into core functionality. Every other platform treats training as a separate system. eLeaP treats it as an integral part of the development record — because regulators and auditors in every industry expect both.
Start Your Free 30-Day Trial. No Credit Card Required
|
Built for Every Regulated Industry ISO 13485 | QMSR / 21 CFR Part 820 |
Industries Served Medical Device & IVD |
For Medical Device Teams
If you are developing medical devices, IVD products, combination products, or SaMD and need Design Controls under ISO 13485 or QMSR, see our dedicated Design Controls System page — purpose-built for FDA and EU MDR compliance.
-
Navigate complex regulatory requirements with confidence — across every major global standard
-
Eliminate the competency gap between development activity and team training — automatically
-
Accelerate development timelines with structured, traceable workflows that eliminate rework
-
Maintain continuous audit readiness without manual documentation effort
Proven Benefits:
-
Faster Regulatory Approvals
Achieve ISO certifications, regulatory approvals, and market clearances faster with automatically generated documentation that meets expectations across ISO 13485, AS9100, IATF 16949, QMSR, ICH, and other major frameworks
-
Reduced Development Costs
Eliminate redundant testing and documentation efforts through clear traceability and reusable templates — so teams spend less time on documentation and more time on development.
-
Improved First-Pass Success
Identify issues early through structured reviews and comprehensive verification protocols, reducing costly late-stage changes, post-market corrections, and re-certification cycles.
-
Streamlined Regulatory Inspections
Provide FDA investigators, notified bodies, certification bodies, and customer auditors with instant access to complete development history, clear traceability, and comprehensive risk documentation — in any regulated industry.
-
Closed-Loop Competency Management
Connect development activity directly to training records. When specifications change, the right people are trained and verified before the next phase begins — automatically, with no manual coordination.
-
Cross-Functional Collaboration
Enable R&D, quality, regulatory, manufacturing, and operations teams to work together efficiently with role-based access and real-time project visibility — across functions, sites, and supply chain partners.
eLeaP’s Design & Development System is trusted by organizations worldwide for its ability to ensure compliance while accelerating development — across medical device, pharmaceutical, aerospace, automotive, food & beverage, and every other regulated industry. Take the next step toward efficient, compliant product development. Start your free trial below or schedule a demo today.
The Design & Development Module is part of the comprehensive eLeaP Quality Management System.