Enterprise Quality Management System (QMS) Software for Regulated Industries
Complete QMS platform with integrated LMS for regulated industries — medical device, pharmaceutical, biotech, manufacturing, aerospace, automotive, food & beverage, and more.
What is eLeaP QMS?
eLeaP is a comprehensive quality management system with enterprise learning management built into one unified platform. Unlike standalone QMS solutions, eLeaP automatically connects quality processes to training verification — closing the compliance gap that exists when QMS and training records live in separate systems.
Complete QMS Modules with Training Integration:
-
Document Control
21 CFR Part 11 compliant document management with version control, approval workflows, and automatic training deployment.
-
Design & Development
Design and development records with full traceability matrix and competency tracking through every phase.
-
Risk Management
ISO 14971, ICH Q9, and FMEA methodologies with risk assessment, mitigation tracking, and residual risk monitoring.
-
Change Control
Complete change lifecycle management with integrated risk assessment and automatic retraining for affected personnel.
-
CAPA Management
Corrective and preventive actions with root cause analysis and effectiveness verification.
-
Event Management
Complaints, nonconformances, and deviations managed end-to-end with configurable workflows.
-
Supplier Management
Vendor qualification, audit management, and certificate tracking linked to your internal quality processes.
-
Training Management
21 CFR Part 11 validated training management with competency assessments and skills tracking.
-
Regulatory Compliance & Standards
Our QMS software supports 21 CFR Part 11 (electronic signatures), QMSR (21 CFR Part 820), ISO 13485:2016, ISO 9001:2015, AS9100, IATF 16949, ICH guidelines, EU MDR, and cGMP requirements. Pre-validated for regulated industries with built-in compliance templates and audit trails.
-
Proven Results
With 20 years serving regulated industries and 1,544 companies served, eLeaP helps medical device, pharmaceutical, biotech, aerospace, automotive, food & beverage, and other regulated organizations achieve faster regulatory submissions, maintain continuous audit readiness, and reduce total quality management costs — all in one integrated platform.
-
Industries Served
Medical Devices, Pharmaceuticals, Biotechnology, Aerospace & Defense, Automotive, Food & Beverage, Digital Health & SaMD, Clinical Research, Regulated Cannabis & Hemp, Cosmetics & Personal Care, Nutraceuticals, Contract Manufacturing (CMO/CDMO), Quality & Regulatory Affairs.
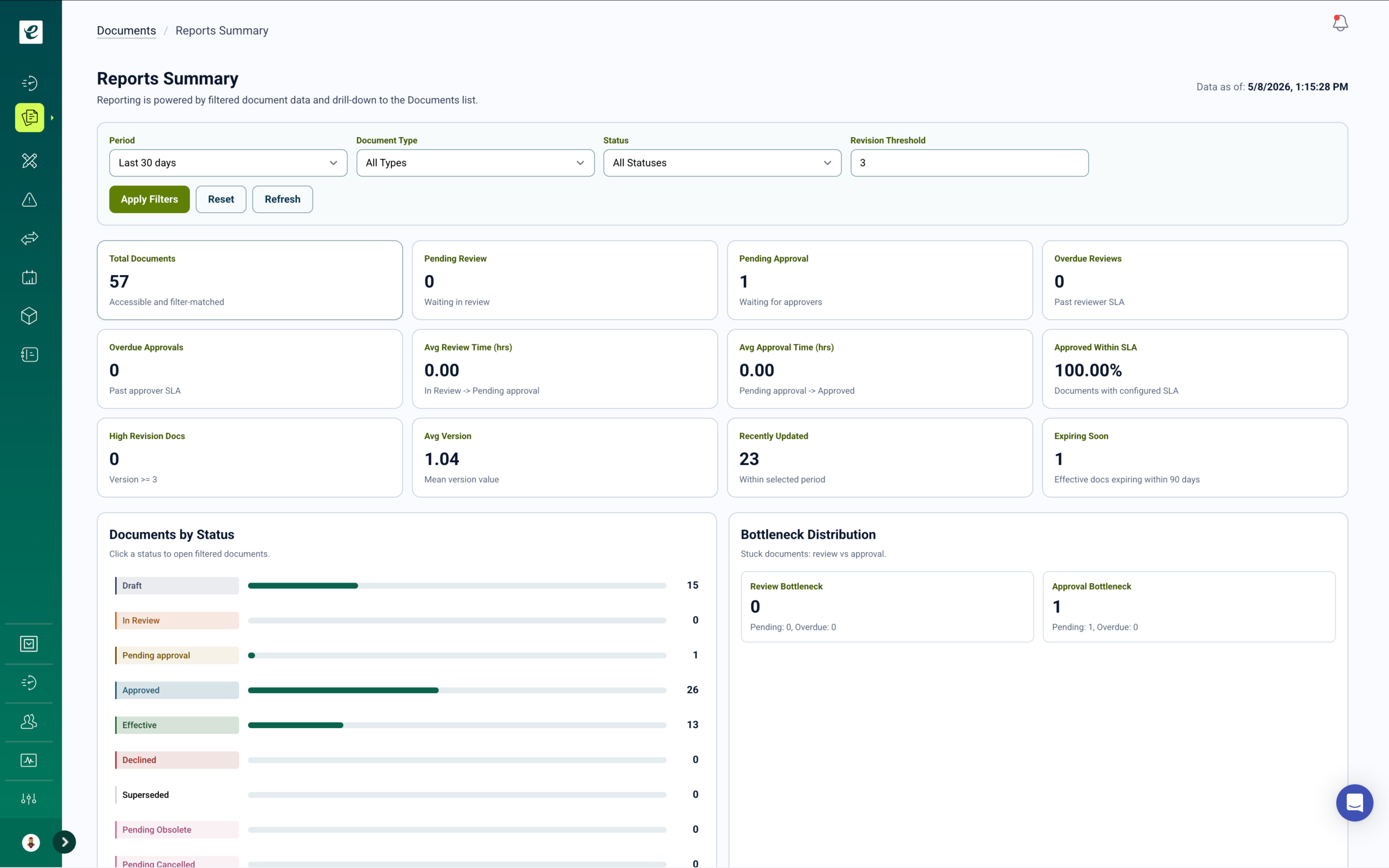
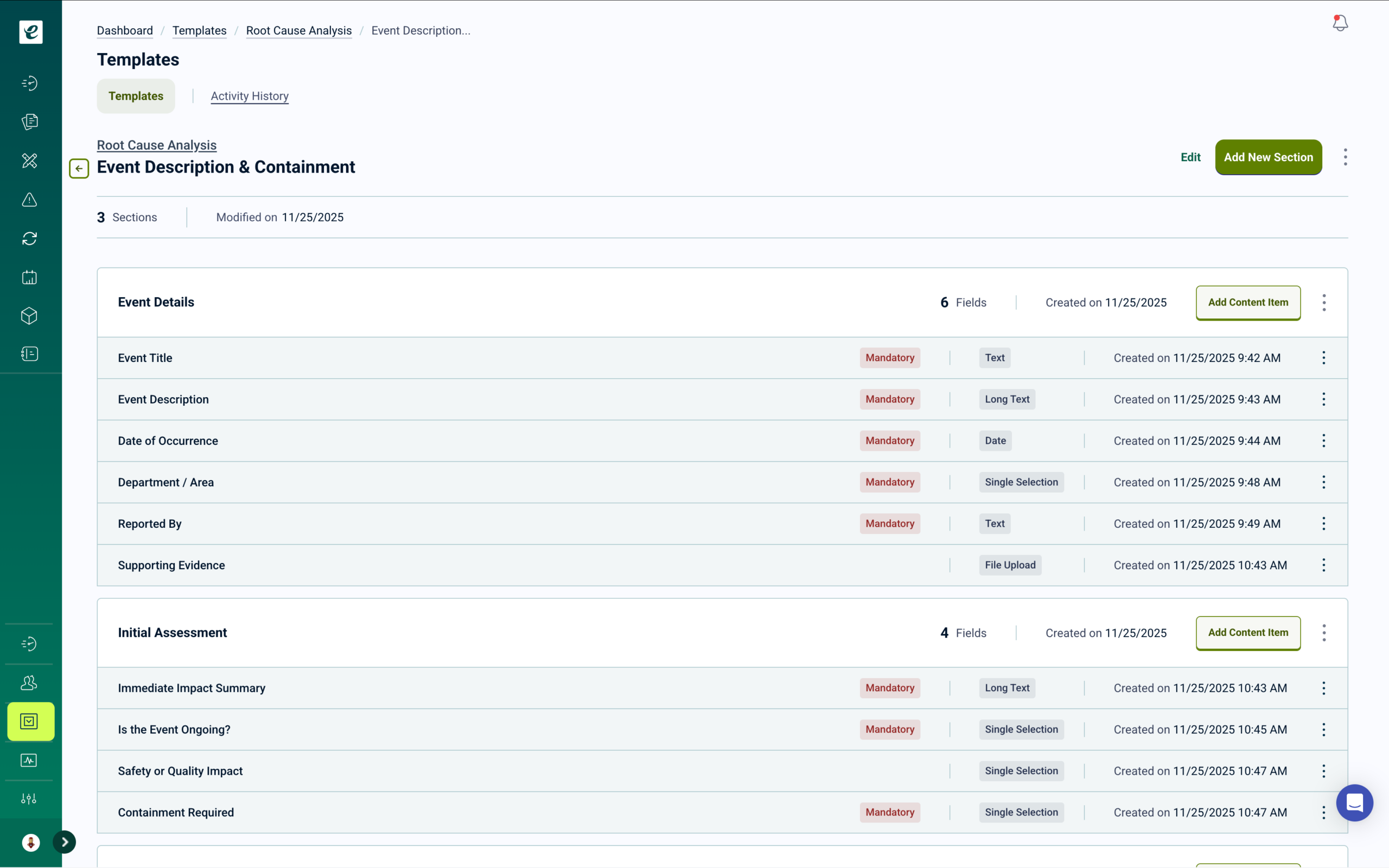
Document Control with Automatic Training Deployment
Manage SOPs, work instructions, forms, and quality procedures with 21 CFR Part 11 compliant document control. Full lifecycle management includes version control, approval workflows, and effective dating. Approved documents automatically become assigned training with completion tracking — so every document revision triggers required retraining for affected personnel. Monitor who needs training, who’s completed it, and maintain comprehensive audit trails across all operational states.
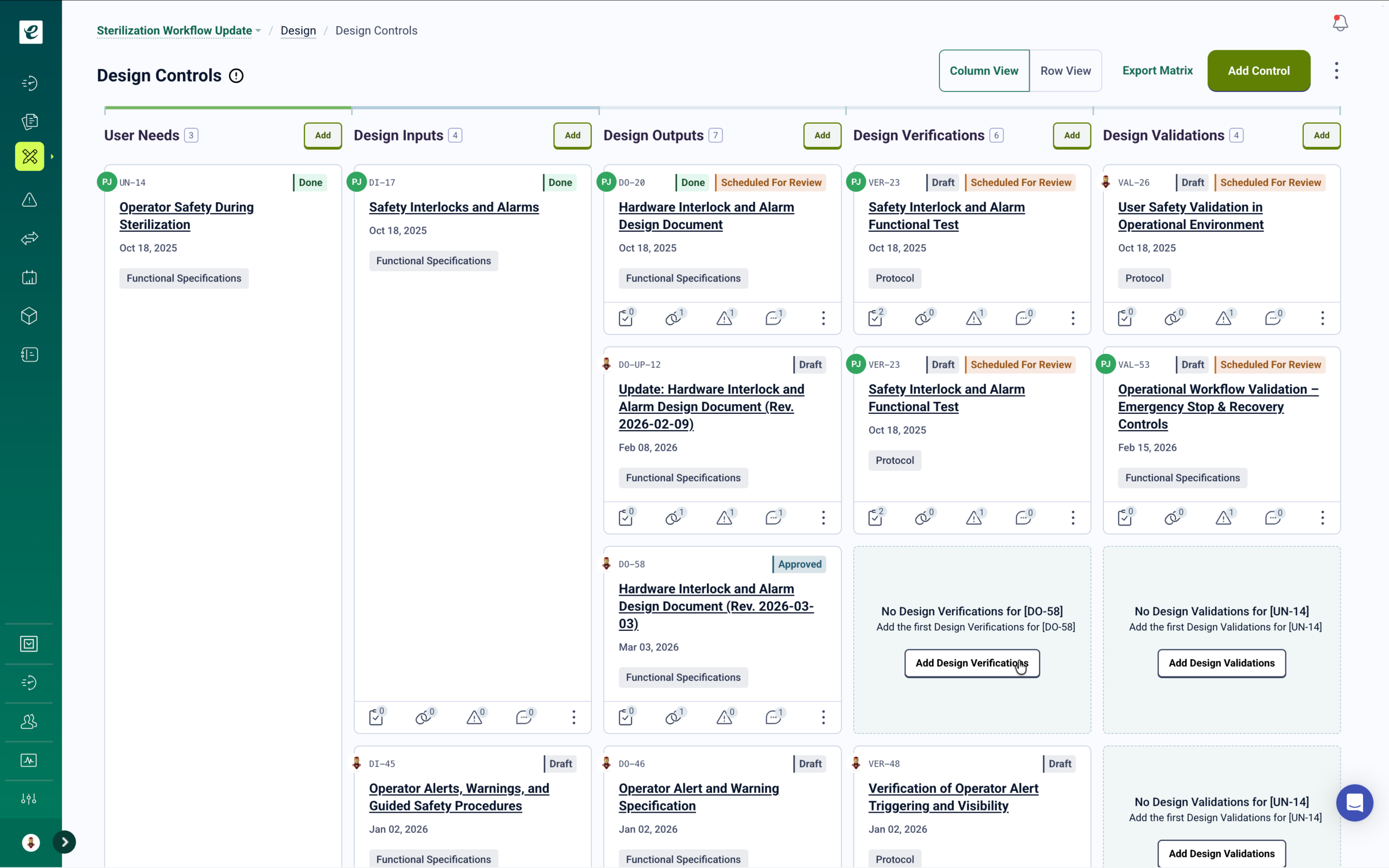
Design & Development with Complete Training Traceability
Manage design and development from concept through commercialization with full traceability across requirements, specifications, verification, and validation. Build comprehensive design and development records including design inputs, outputs, reviews, and transfer documentation. Training requirements cascade automatically through development phases — when specifications change, affected engineers and technicians are notified and retrained. Generate audit-ready documentation with integrated competency records for faster regulatory approvals and certification audits.
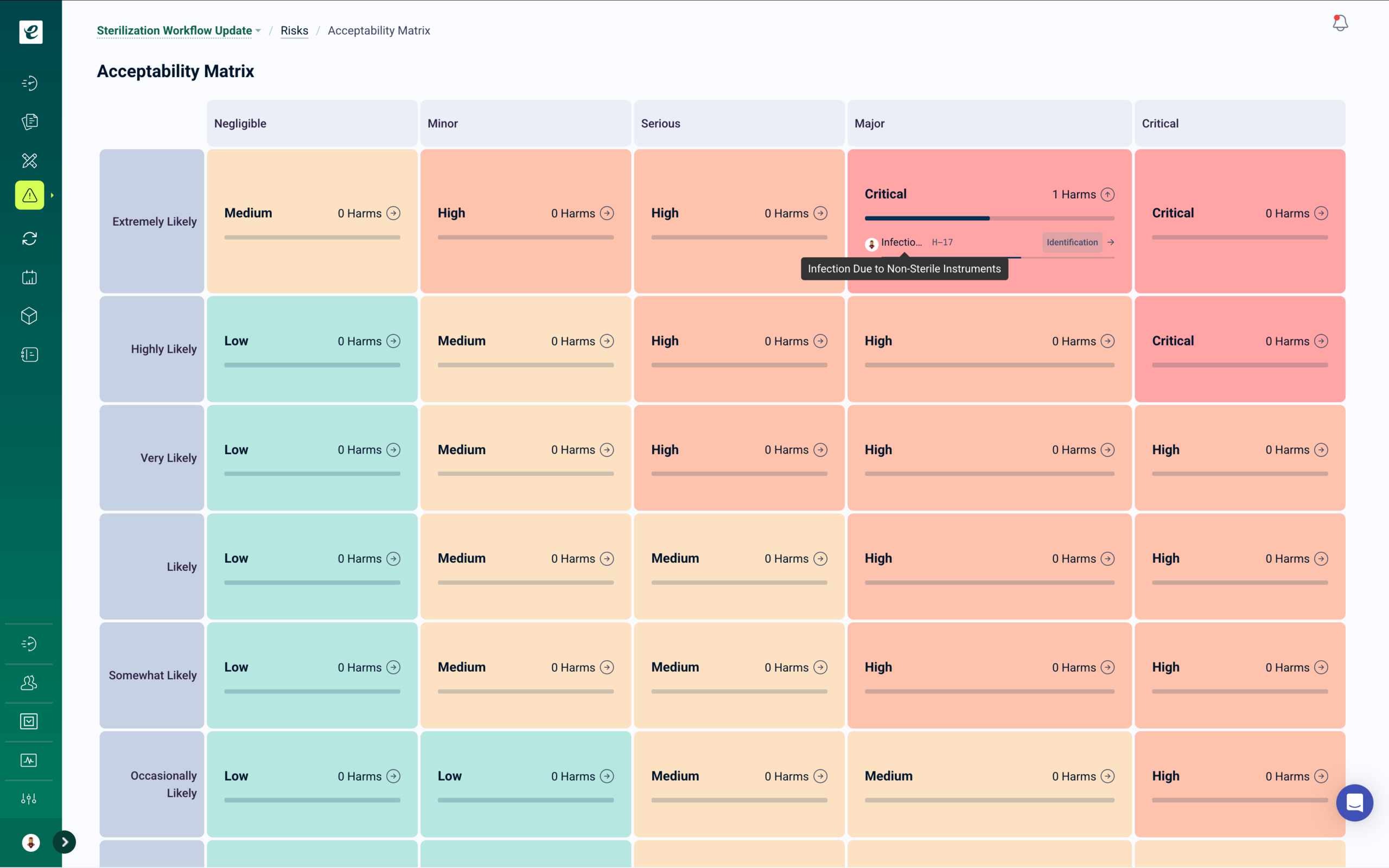
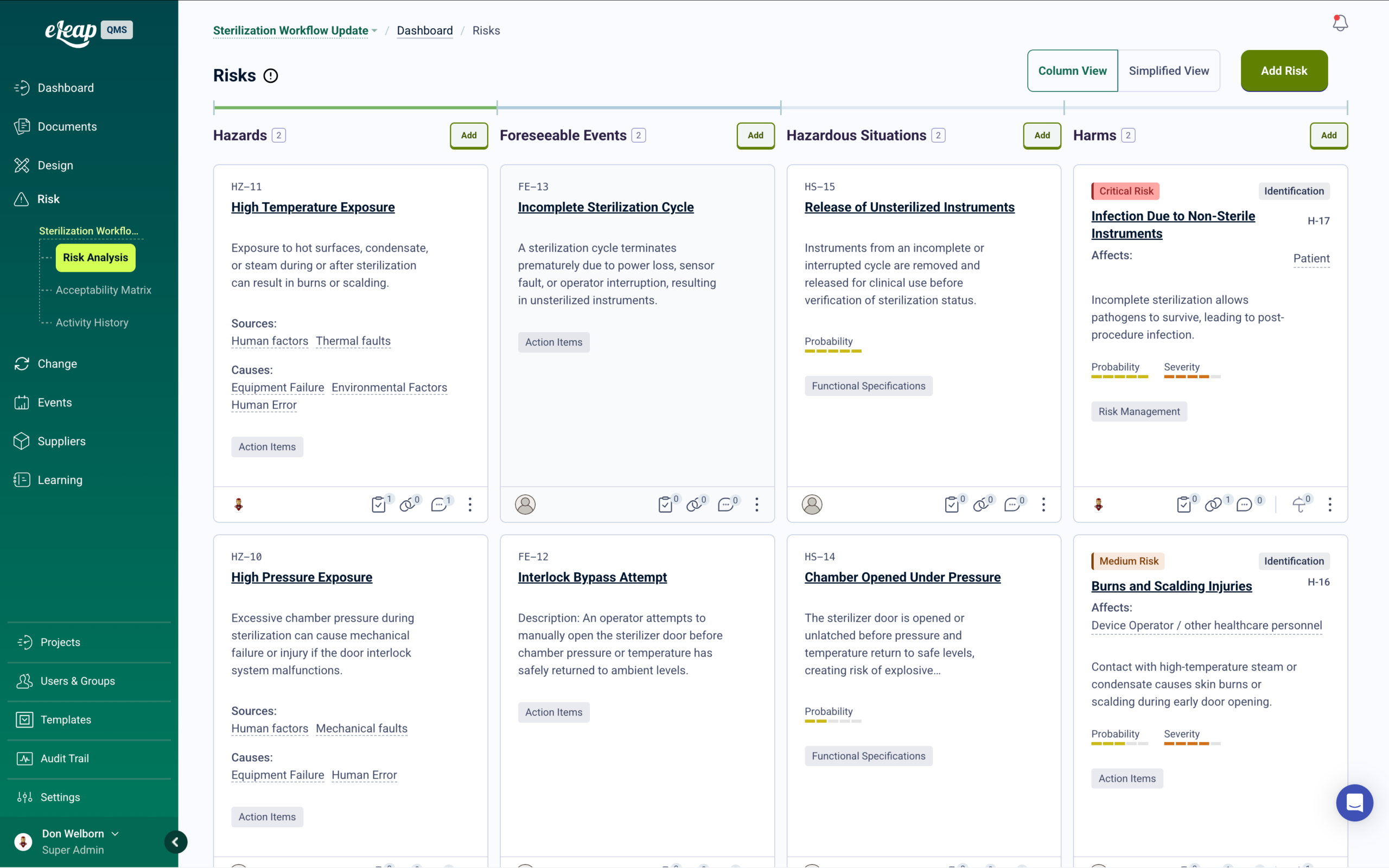
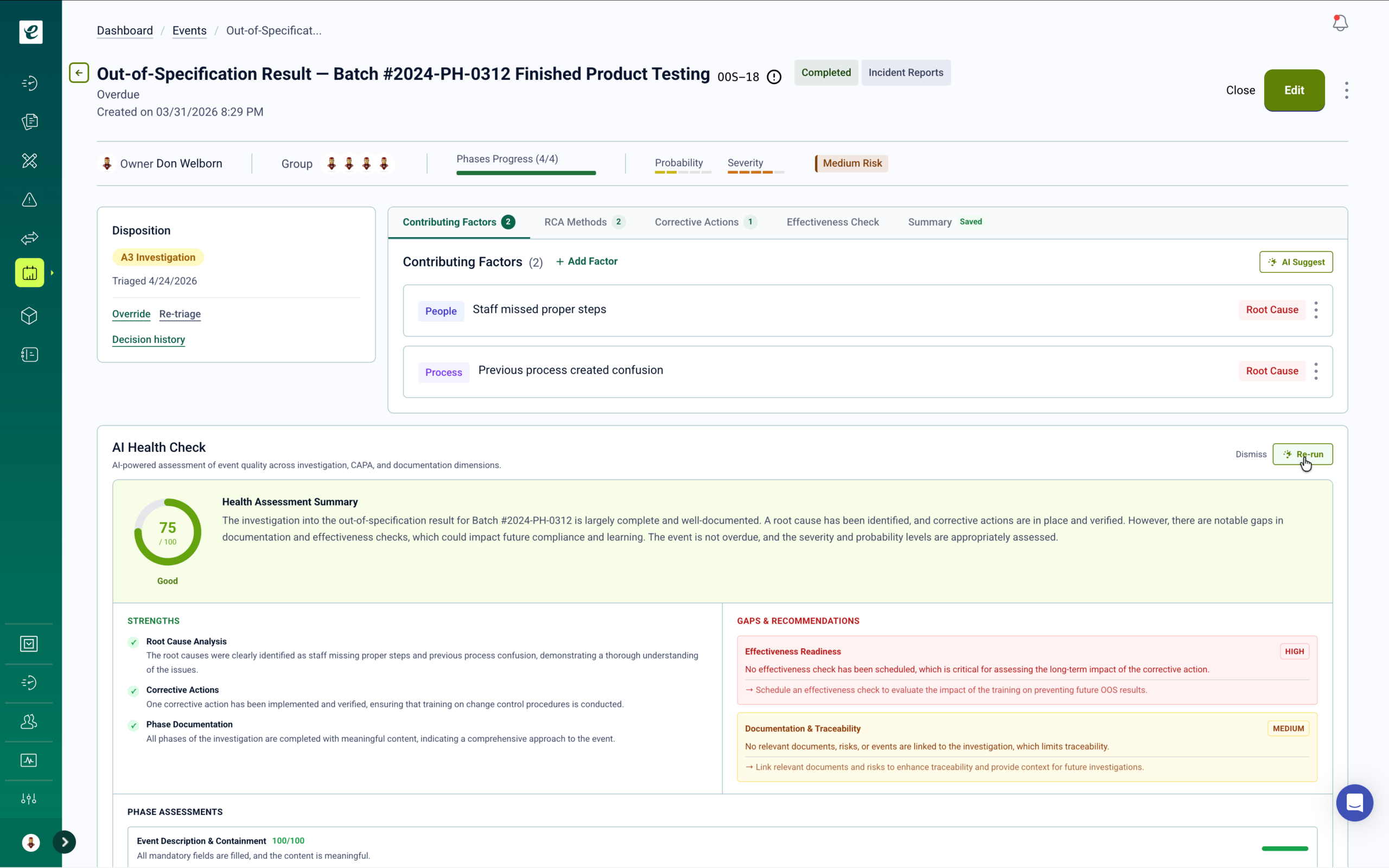
Integrated Risk Management with Automated Mitigation Training
eLeaP supports ISO 14971 (MedTech), ICH Q9 (Pharma), and FMEA methodologies in one unified platform. Conduct comprehensive assessments using customizable probability and severity matrices, generate risk priority numbers, and track residual risks in real-time. When new control measures are identified, targeted training is automatically deployed to affected personnel.
Visualize Paths to Harm, maintain living risk registers, and ensure every mitigation strategy includes verifiable proof of competency for your next audit or inspection.
Adaptive Event Management with Custom Workflow Templates
Stop forcing your quality processes into rigid software silos. eLeaP’s powerful customizable templates allow you to build tailored workflows for CAPAs, NCRs, deviations, RCA investigations, and any other quality event unique to your industry. From initiation to closure, you control the data fields, approval logic, and containment steps to match your specific SOPs.
Integrated Operations: Change, Suppliers, Templates & Training.
eLeaP centralizes the lifecycle of your most critical quality operations — Change Control for document and process modifications, Supplier Management for qualification, audits, and quality agreements, and Customizable Templates for building industry-specific workflows for any quality event.
These modules are natively linked to the integrated Training Management engine, ensuring that any operational shift — whether a document change or a supplier update — automatically triggers targeted retraining and competency tracking for affected teams, maintaining a continuous state of audit readiness.
One Platform for Compliance, Integration, and Audit Readiness.
eLeaP delivers a comprehensive QMS with seven core modules — documents, design & development, risk, change, quality events, suppliers, and learning — operating as one connected system. Training is automated across every module, triggering retraining based on document approvals, design changes, quality events, and audits, so your team stays compliant without manual coordination.
-
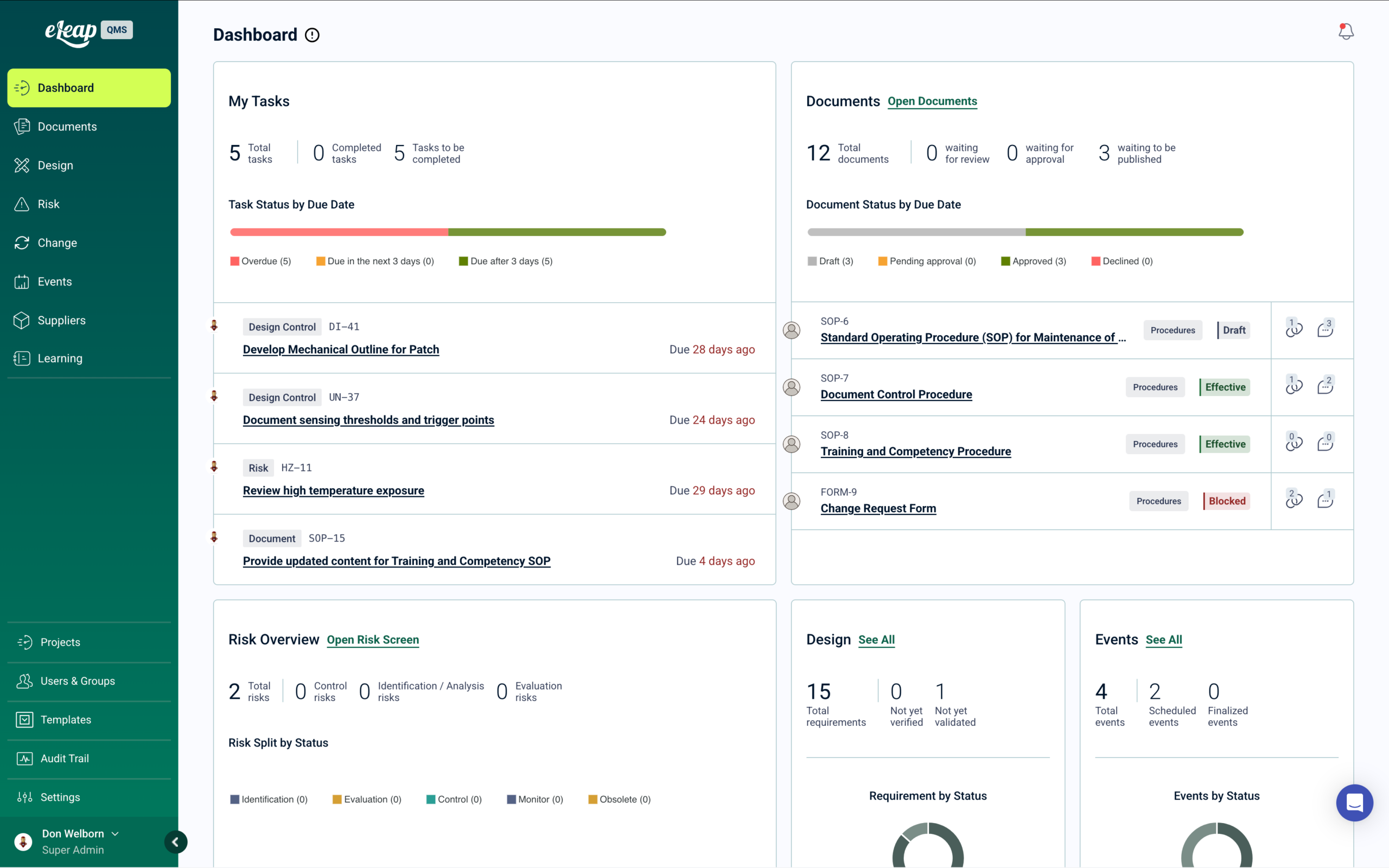
See Your Complete Quality Status
Real-time visibility across documents, design & development, risk, change, events, suppliers, and learning. Every quality action is linked to training. One platform. One sign-on. One source of truth.
-
Compliance Without Manual Work
Approved documents trigger training. Design & development, risk, and change updates cascade requirements. Events and audits close gaps. Learning tracks completion automatically — no manual handoffs.
-
Connected Data Drives Better Decisions
All seven systems — Documents, Design & Development, Risk, Change, Events, Suppliers, Learning — operate as one integrated QMS. Root cause analysis links training and performance data to every audit trail.
Trusted by Regulated Industries for 20+ Years
Medical device, pharmaceutical, biotechnology, aerospace, automotive, food & beverage, and other regulated industries trust eLeaP to manage compliance at scale. Built on 20 years of proven infrastructure, our platform embeds compliance into daily workflows — from manufacturing and clinical research to contract operations (CMO/CDMO) and beyond. Fully 21 CFR Part 11 compliant with complete audit trails and validated documentation — so every team can focus on quality improvement while the system ensures no requirement is overlooked.
The eLeaP system is very user-friendly, and it significantly improved our current training process. The thing that was most impressive to me was the customer service.