QMS for Manufacturing
The only eQMS that unifies quality management processes and compliance training in one integrated system — for regulated manufacturers.
Quality Management System for Regulated Manufacturing Operations
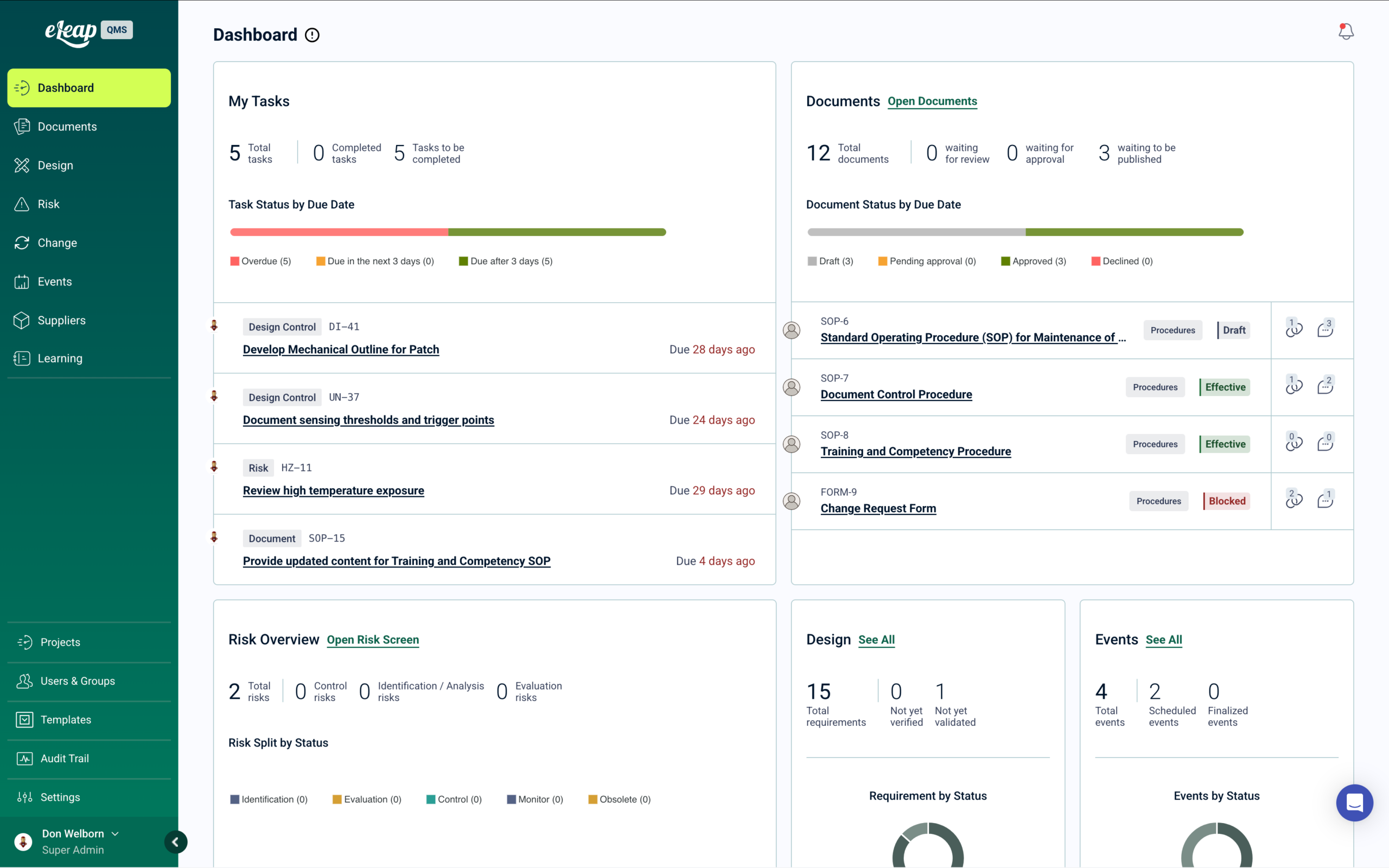
eLeaP is the only Quality Management System with enterprise Learning Management built in — designed for regulated manufacturers operating under ISO 9001, IATF 16949, and customer-specific quality requirements. Where other platforms require a separate LMS, eLeaP automates personnel qualification across every quality process from a single validated system. → Get a Demo.
How It Works:
When work instructions or quality procedures are approved, they automatically become training assignments with completion tracking. Process changes cascade required training to affected production roles. Risk assessments trigger training when new control measures are implemented. CAPAs and nonconformance investigations drive targeted corrective training with documented effectiveness verification — all traceable for ISO certification audits and customer source inspections.
Our platform deploys in weeks, with templates aligned to ISO 9001:2015 and IATF 16949 requirements. Trusted by contract manufacturers, component suppliers, and regulated manufacturers across automotive, industrial, defense, and general manufacturing sectors.
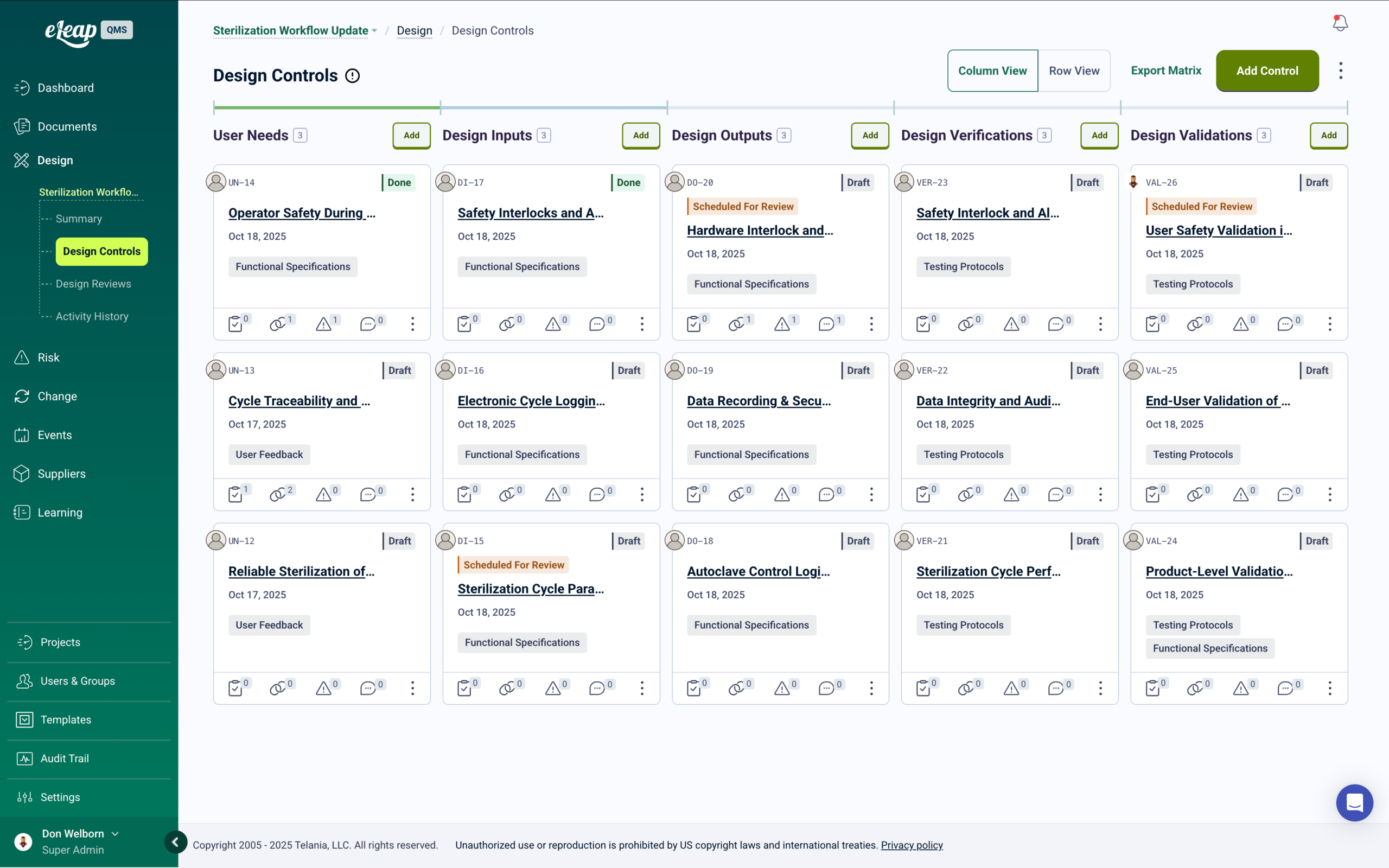
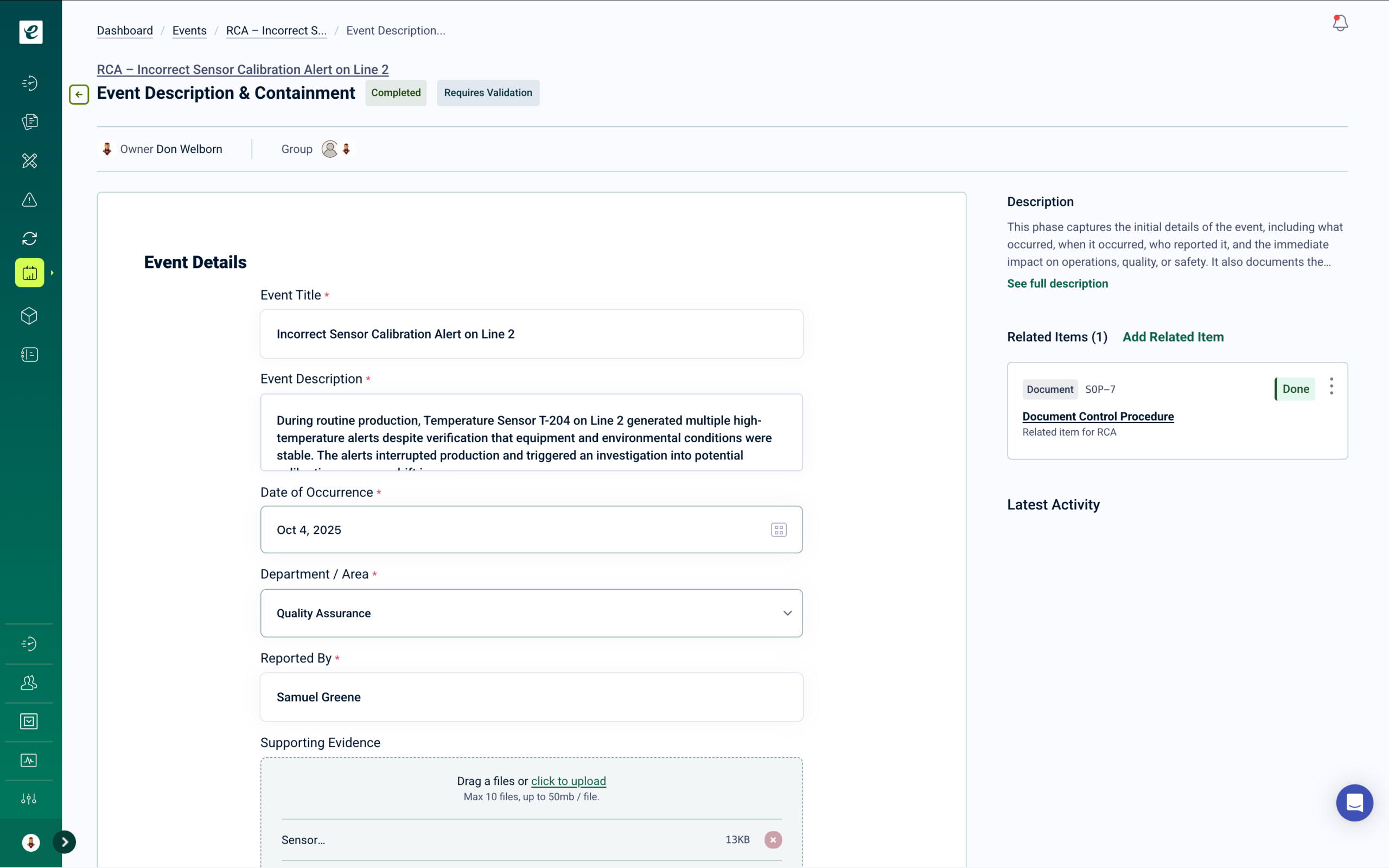
ISO 9001 Document Control and Records Management
Controlled procedures, work instructions, quality plans, inspection records, and customer-required documentation — managed in one audit-ready system with version control and automatic training deployment on approval.
Document Control Built for ISO 9001 Compliance
ISO 9001:2015 requires documented information to be controlled to ensure it is available, suitable for use, and adequately protected. Manufacturers manage large volumes of document types — quality manuals, process procedures, work instructions, inspection forms, and customer-specific quality requirements. eLeaP controls the full document lifecycle with version-controlled workflows, electronic approval signatures, and complete audit trails.
When a document is approved and released, training automatically deploys to every affected role. Version control prevents use of superseded work instructions on the production floor. When a certification auditor or customer quality representative requests documentation records, everything is in one place, versioned, and immediately available. Learn more. Get a Demo.
Design and Development Controls with APQP Support
Design inputs, outputs, reviews, verification, and validation records — including APQP phase management, PPAP documentation, and design FMEA — with full traceability from requirements through production approval.
Design Controls Aligned to ISO 9001 and IATF 16949
ISO 9001:2015 and IATF 16949 require structured design and development processes with documented planning, inputs, outputs, reviews, and validation. Advanced Product Quality Planning (APQP) provides the framework for phased development with customer alignment, and the Production Part Approval Process (PPAP) provides evidence that production processes can consistently produce conforming product. eLeaP manages design control documentation through all phases with complete traceability.
As design phases progress, training requirements automatically adjust for engineering, production, and quality personnel. Design changes cascade training to impacted teams before implementation. Design reviews create verification records. Generate design history documentation with complete training records and approval evidence for customer PPAP submissions and ISO audits. Get a Demo.
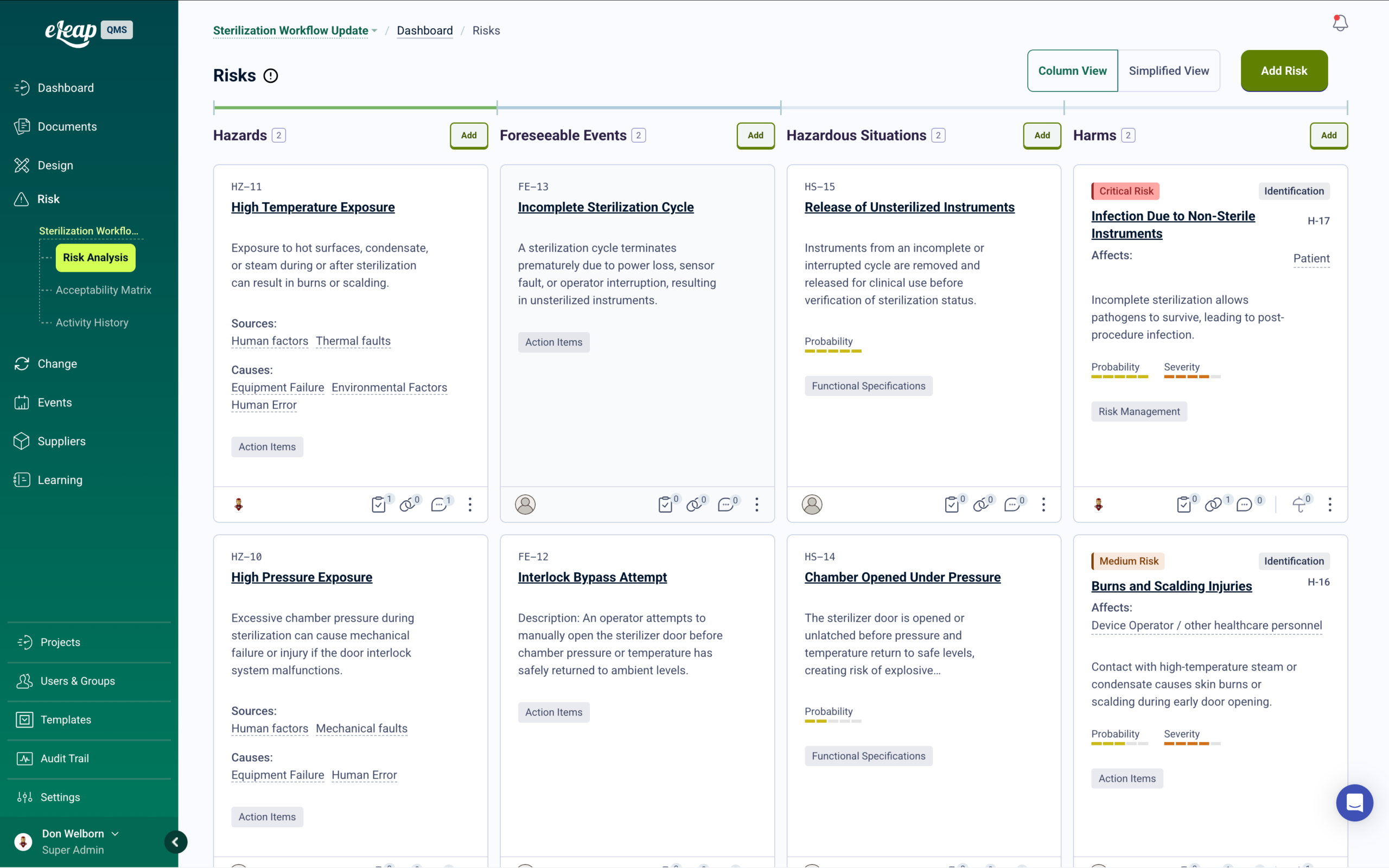
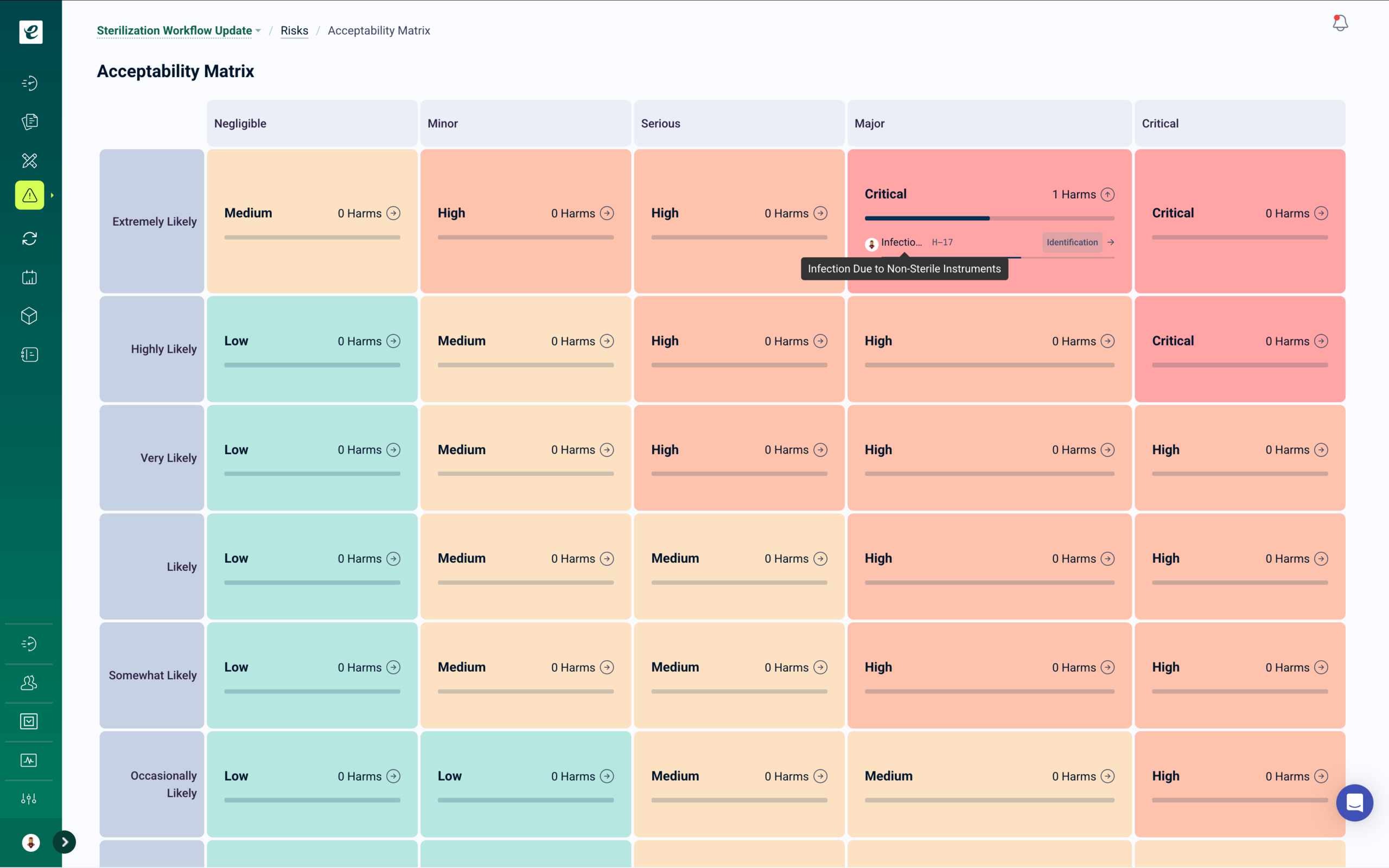
Risk Management with Integrated Training
Process failure mode analysis, operational risk assessments, and quality risk management — with automatic training deployment when control measures are introduced or updated.
When risk assessments identify new control measures, training automatically deploys to affected personnel. Root cause analysis connects quality events to training history, identifying whether issues stem from knowledge gaps, process failures, or tooling problems. Risk reviews, control effectiveness monitoring, and CAPA linkage are maintained in one traceable system ready for certification audits.
Risk-Based Thinking for Manufacturing Quality Operations
ISO 9001:2015 requires organizations to consider risks and opportunities when planning and implementing the quality management system. IATF 16949 goes further, requiring process FMEA, product FMEA, and control plans linked to risk mitigation. eLeaP’s risk management module supports risk assessments with probability and severity scoring, process FMEA documentation, and control plan linkage. Get a Demo.
Change Control with Automatic Training
Engineering changes, process modifications, equipment updates, and procedure revisions automatically trigger training for affected personnel with escalation workflows and verified implementation.
Manufacturing operations require rigorous change control to maintain process capability and product conformance. ISO 9001 and IATF 16949 require controlled processes for changes that could affect product quality or process parameters. eLeaP manages change requests through impact assessment, approval, implementation, and verification with automated training deployment built into the workflow.
Production Change Control That Keeps Qualification Current
When a change is approved, training deploys immediately to all roles identified in the impact assessment. For customer-directed changes, traceability from change request through training completion and implementation verification provides the evidence required for customer notification and approval. Change closure requires documented training completion and competency verification. Get a Demo.
CAPA, NCR, and Nonconformance Management
Nonconformance reports, corrective action requests, customer complaints, and audit findings automatically identify training needs and deploy assignments — connecting investigations, corrective actions, and personnel competency in one traceable system.
Nonconformance Management Built for Manufacturing Quality
ISO 9001:2015 requires organizations to take action to control and correct nonconforming outputs and to deal with the consequences. IATF 16949 adds specific requirements for escalation, customer notification, and field action processes. eLeaP’s events management module handles NCRs, CAPAs, customer complaints, and audit findings with structured workflows and automatic training deployment when root cause analysis identifies competency gaps.
When an NCR identifies a procedural error or audit finding points to inadequate operator qualification, training assignments deploy automatically. CAPA closure requires documented training completion and effectiveness verification. Customer 8D responses and corrective action evidence include complete training records. All records are maintained and traceable for ISO certification audit. Get a Demo.
Supplier Quality Management for Manufacturing Supply Chains
Approved supplier list management, supplier qualification, performance monitoring, and audit management — with training triggers when supplier issues identify internal process gaps.
When supplier audits identify gaps in internal receiving, inspection, or qualification procedures, training automatically deploys to affected internal teams. Supplier performance metrics, audit findings, and corrective action records are tracked in a system directly linked to your broader quality operations.
ISO 9001 and IATF 16949 Supply Chain Quality Management
ISO 9001:2015 requires organizations to determine and apply criteria for evaluating, selecting, monitoring, and re-evaluating external providers. IATF 16949 adds specific requirements for supplier development, second-party audits, and supplier quality management system requirements. eLeaP’s supplier management module supports approved supplier list management, quality agreement documentation, incoming inspection records, and supplier audit management. Get a Demo.
ISO 9001 Training Compliance — Built In, Not Added On
Enterprise learning management is native to the platform. ISO 9001 competency requirements, role-specific qualification, and effectiveness verification are managed automatically — not tracked in a separate system.
ISO 9001:2015 clause 7.2 requires organizations to determine the necessary competence for persons doing work that affects quality, provide training or other actions to achieve competency, evaluate the effectiveness of actions taken, and retain documented information as evidence of competency. Meeting these requirements across production, engineering, inspection, and quality roles — with changing procedures and ongoing quality events — requires more than a training system that integrates with your QMS.
eLeaP’s learning management is native to the platform. Documents approved in Document Management automatically create training assignments. CAPAs closed in Events trigger effectiveness verification. Change Control approvals deploy role-based training before implementation. Qualification matrices show competency status across all roles in real time. When a certification auditor asks for training records and effectiveness evidence, you answer immediately — from one system.
Building ISO 9001 Compliance Into Every Workflow
ISO 9001 certification audits and customer source inspections consistently review training records, nonconformance investigations, and the traceability between quality events and corrective actions. eLeaP makes training an automatic output of every quality process.
-
Automatic Training Triggers
Procedure approvals, process changes, NCR closures, and CAPA actions automatically deploy training to the right personnel at the right time.
-
Real-Time Qualification Verification
Competency status is visible across all roles and production lines — no spreadsheets, no manual tracking.
-
Inspection-Ready by Design
Complete audit trails connect quality decisions to training records. When certification auditors and customer quality representatives ask, you answer immediately.
Quality and Training Working as One System
Manufacturing quality management should drive operational excellence — not administrative burden. eLeaP gives quality teams the tools to maintain certification compliance while keeping production teams qualified and informed. Get a Demo.
-
Continuous Training Integration
Every quality process — documents, design, risk, change, events, suppliers — automatically drives training when it matters.
-
Connected Quality Data
All seven modules work as one system. No disconnected records. No manual handoffs. Complete audit trails.
-
Competency-Driven. Root Cause Analysis
Investigations connect training history, competency assessments, and quality outcomes — identifying whether issues stem from knowledge gaps or process failures.
-
Here, When You Need Us
Our quality experts support ISO 9001 and IATF 16949 implementation, certification preparation, and continuous improvement programs.