The Document Management System Built for Regulated Industries
eLeaP manages the full document lifecycle — version control, approval workflows, and linked training — so your team stays compliant without the manual work.
The Document Management Problem: Book a Demo
- ❌ Employees lose 2.5 hours daily — 12+ hours weekly — just searching for documents and current versions.
- ❌ Manual approval routing often delays critical SOP updates by weeks, leaving teams working on outdated procedures.
- ❌ Regulatory observations frequently cite inadequate document control as a primary finding.
- ❌ Without a centralized system, teams face a chaotic rush to locate current documents and training records.
- ❌ Disconnected systems mean employees are often not trained on the latest revisions, creating “root cause” for compliance failures.
The eLeaP Solution:
- ✅ Automate version control and administrative tracking, allowing your quality team to focus on high-value improvements rather than manual paperwork.
- ✅ Unlike standalone systems, eLeaP automatically triggers training assignments of controlled documents, ensuring 100% of your team is trained on the latest procedures.
- ✅ Use parallel routing and intelligent automated escalations to move SOPs from draft to “Effective” status in days, not weeks.
- ✅ Ensure full compliance with 21 CFR Part 11, EU Annex 11, and GAMP 5 through built-in electronic signatures and indelible audit trails.
- ✅ Impress inspectors with instant retrieval of any document’s full history, including linked training records and change controls.
Stop risking compliance violations with inadequate document control. The Document Management module provides a comprehensive system that automates version control, approval workflows, and training assignments while meeting FDA, EU, and ISO requirements. See How eLeaP Manages Compliance Docs → Free Demo.
Unlike standalone document systems that create data silos, eLeaP integrates document management with training records to ensure every employee is automatically trained on the latest procedures. Start your free 30-day trial. No credit card required. Get full feature access and implementation support. Or schedule a free session with one of our solution experts.
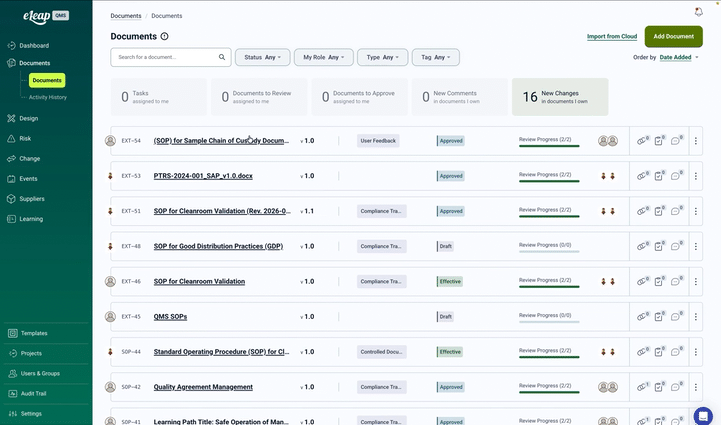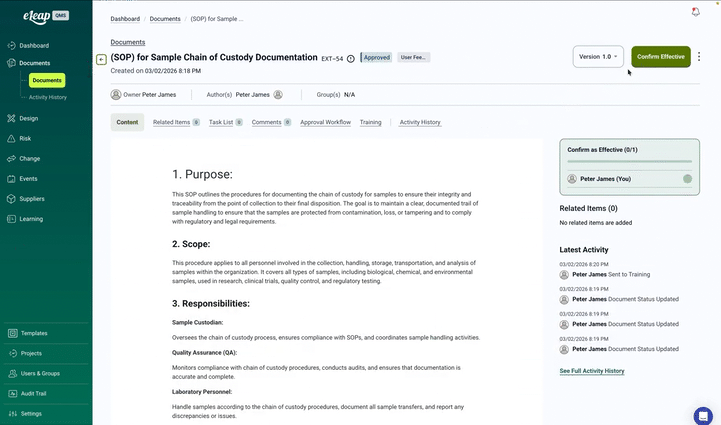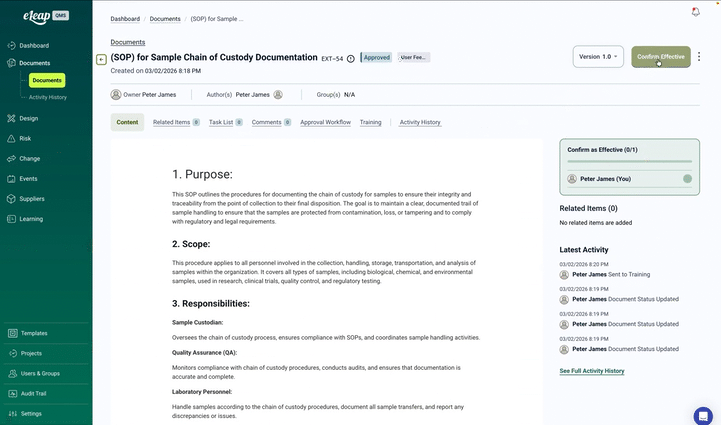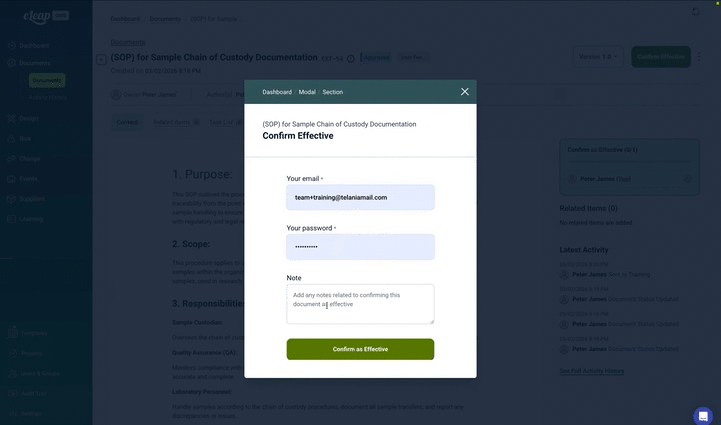
Transforming Document Control for Regulated Industries
Whether you’re developing medical devices, manufacturing pharmaceuticals, or producing diagnostics, document control is the foundation of your quality system. Traditional approaches using shared drives, paper systems, or disconnected tools put companies at risk for compliance failures and operational inefficiencies. eLeaP’s Document Management System delivers enterprise-grade control without enterprise complexity.
At the core of eLeaP Quality Management Software is its powerful Document Management Module, a comprehensive system offering complete lifecycle control from creation through obsolescence. With automated version control, compliant electronic signatures, and seamless training integration, eLeaP ensures your documentation meets the strictest regulatory requirements across all major markets.
Key Features
-
Intelligent Document Lifecycle Management
Control documents from draft through obsolescence with configurable workflows, automatic versioning, and clear effective dates. Maintain complete revision history for any document type—SOPs, work instructions, forms, or specifications.
-
Regulatory-Compliant Electronic Signatures
Built-in e-signatures meet 21 CFR Part 11, EU Annex 11, and GAMP 5 requirements with authentication, audit trails, and signature manifestations. Configure signature meanings and approval sequences by document type.
-
Flexible Approval Workflows
Design multi-level approval chains with parallel and sequential routing options. Set automatic escalations, out-of-office delegates, and track bottlenecks with real-time workflow dashboards.
-
Automated Training Integration
Trigger training assignments automatically when documents change. Track read-and-understood acknowledgments, comprehension assessments, and effectiveness checks. Maintain complete training records linked to document versions.
-
Advanced Search and Retrieval
Find any document instantly with full-text search, metadata filtering, and custom tags. Create document collections for projects, audits, or regulatory submissions. Export packages with all supporting documentation.
-
Controlled Access and Distribution
Implement role-based permissions with granular control over viewing, editing, and approving. Manage external distribution with watermarking, expiration dates, and access logs.
-
Quality Event Integration
Link documents directly to change controls, CAPAs, deviations, and complaints. Maintain bi-directional traceability between quality events and documentation updates.
-
Global Compliance Support
Meet requirements for FDA (21 CFR Parts 11, 211, 820), EU MDR/IVDR, ISO (9001, 13485, 15189), ICH guidelines, and country-specific regulations with configurable compliance rules.
Why Select eLeaP Quality?
With over 20 years serving regulated industries, eLeaP understands that one size doesn’t fit all. Our Document Management module adapts to your specific regulatory requirements, whether you’re a medical device startup, pharmaceutical manufacturer, clinical laboratory, or biotechnology company. The integrated QMS+LMS platform eliminates gaps between document control and training—the root cause of many compliance failures.
-
Meet FDA, EU, and ISO requirements from day one
-
Reduce document control overhead by 80%
-
Ensure 100% training compliance automatically
-
Scale from startup to enterprise without switching systems
Proven Benefits:
-
Accelerated Product Development
Reduce document approval cycles from weeks to days with automated routing and parallel reviews. Get products to market faster without compromising compliance.
-
Reduced Compliance Risk
Eliminate observations and warning letters related to document control. Maintain inspection-ready documentation with complete audit trails and training records.
-
Lower Operational Costs
Cut document management overhead while reducing errors that lead to deviations, investigations, and corrective actions. Free quality teams to focus on value-added activities.
-
Successful Regulatory Inspections
Impress inspectors with instant document retrieval, clear revision histories, and automated training matrices. Generate compliance reports in minutes, not days.
-
Improved Collaboration
Enable cross-functional teams to work together efficiently with commenting tools, version comparison, and automated notifications for document changes.
-
Flexible Scalability
Start with essential documents and scale to thousands without performance issues. Add sites, divisions, or acquisitions without system constraints.
The eLeaP Document Management Module is trusted by life sciences companies worldwide for its ability to ensure compliance while reducing administrative burden. Take the next step toward efficient, compliant document control. Start your free trial below or schedule a demo today.
The Document Management Module is part of the comprehensive eLeaP Quality Management System.