Quality Management System (QMS) for Regulated Industries
The Only eQMS Platform That Unifies Quality Processes and Training Compliance in One Integrated System

Enterprise Quality Management System for Regulated Industries
eLeaP is the only Quality Management System with Enterprise Learning Management built into core QMS functionality. Quality events, document approvals, design & development changes, and risk assessments automatically deploy the right training to the right people — closing the compliance gap between quality processes and training records. → Get a Free Demo or Start a Free Trial.
How It Works:
When SOPs are approved, they automatically become training assignments with completion tracking. Design and development changes cascade required training to affected teams. Risk assessments trigger mitigation training for identified hazards. Change orders prompt retraining for impacted personnel. CAPAs and audit findings drive targeted training assignments with effectiveness verification.
Proven Results:
Our pre-validated platform includes built-in 21 CFR Part 11 compliance and templates for FDA, ISO, and ICH requirements — so implementation is faster and audit readiness is built in from day one. Trusted by organizations across medical device, pharmaceutical, biotechnology, aerospace, automotive, food & beverage, clinical research, cannabis, cosmetics, nutraceuticals, contract manufacturing, and other regulated industries worldwide.
Document Control with Integrated Training
Full document lifecycle management with automatic training deployment, completion tracking, and effectiveness measurement.
Enterprise Document Management Built for Quality Systems
Control your entire document lifecycle — from draft through obsolescence — with 21 CFR Part 11 compliant electronic signatures and audit trails. Manage SOPs, work instructions, forms, specifications, and policies with version control and automated approval workflows.
When documents are approved, they automatically become training assignments for affected roles. Track read-and-understood acknowledgments, test comprehension, and maintain complete training records — all within the same system. Learn more. Learn more.

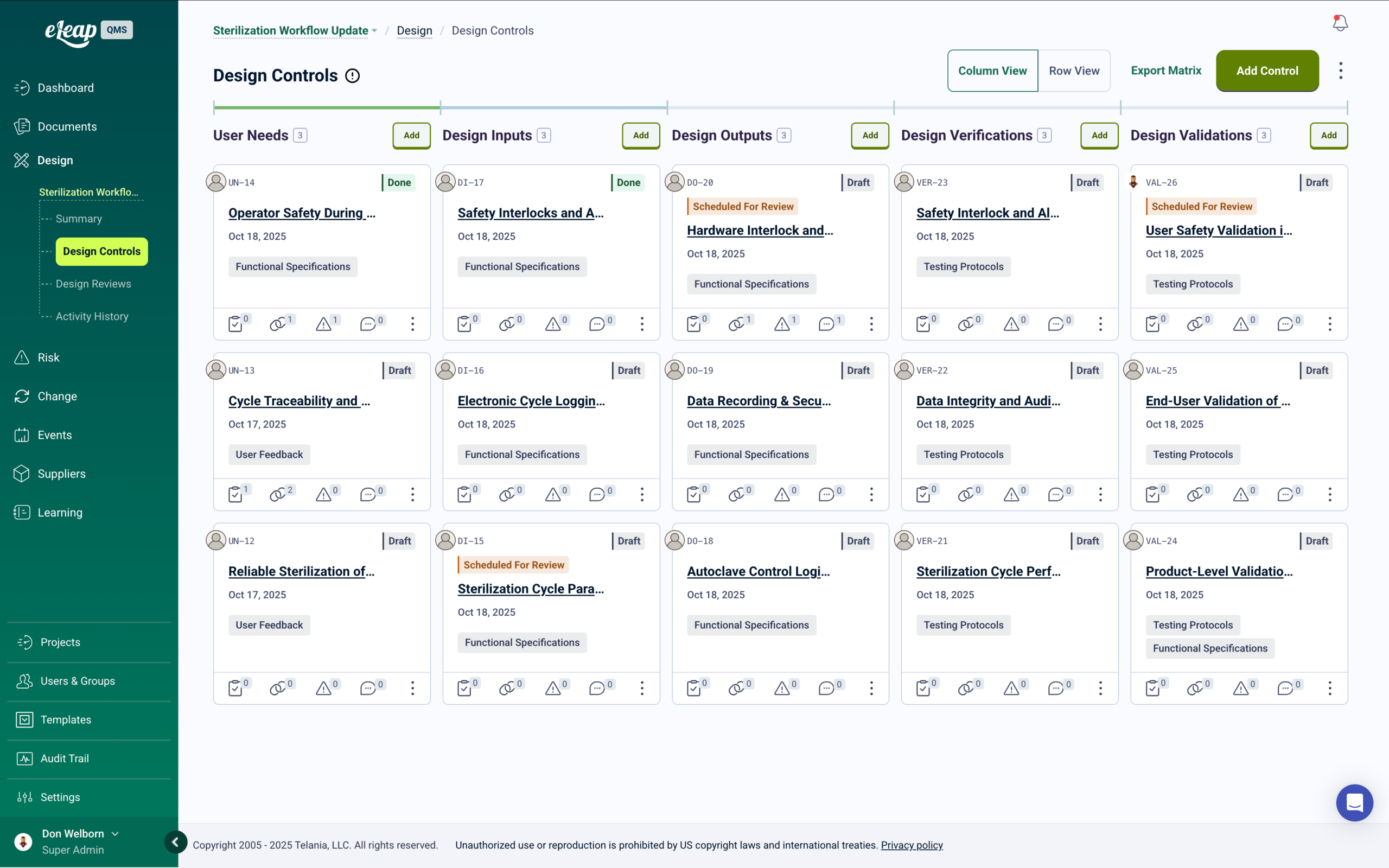
Design & Development with Built-In Competency Management
As design and development requirements progress through each phase, training automatically cascades to affected teams — with full traceability from inputs through validation.
Your design and development process is unique. eLeaP’s integrated Design & Development module manages requirements from inputs through verification and validation with full traceability across every phase — creating and controlling specifications as they progress through each development stage.
The Foundation for Product Quality and Team Competency.
As specifications advance through each phase, eLeaP automatically identifies who needs training and deploys it — no manual coordination required. Design reviews generate verification records. Development changes trigger immediate retraining for affected teams. Every approval, competency assessment, and training record is captured and linked — so when an auditor asks, everything is already there. Learn more.
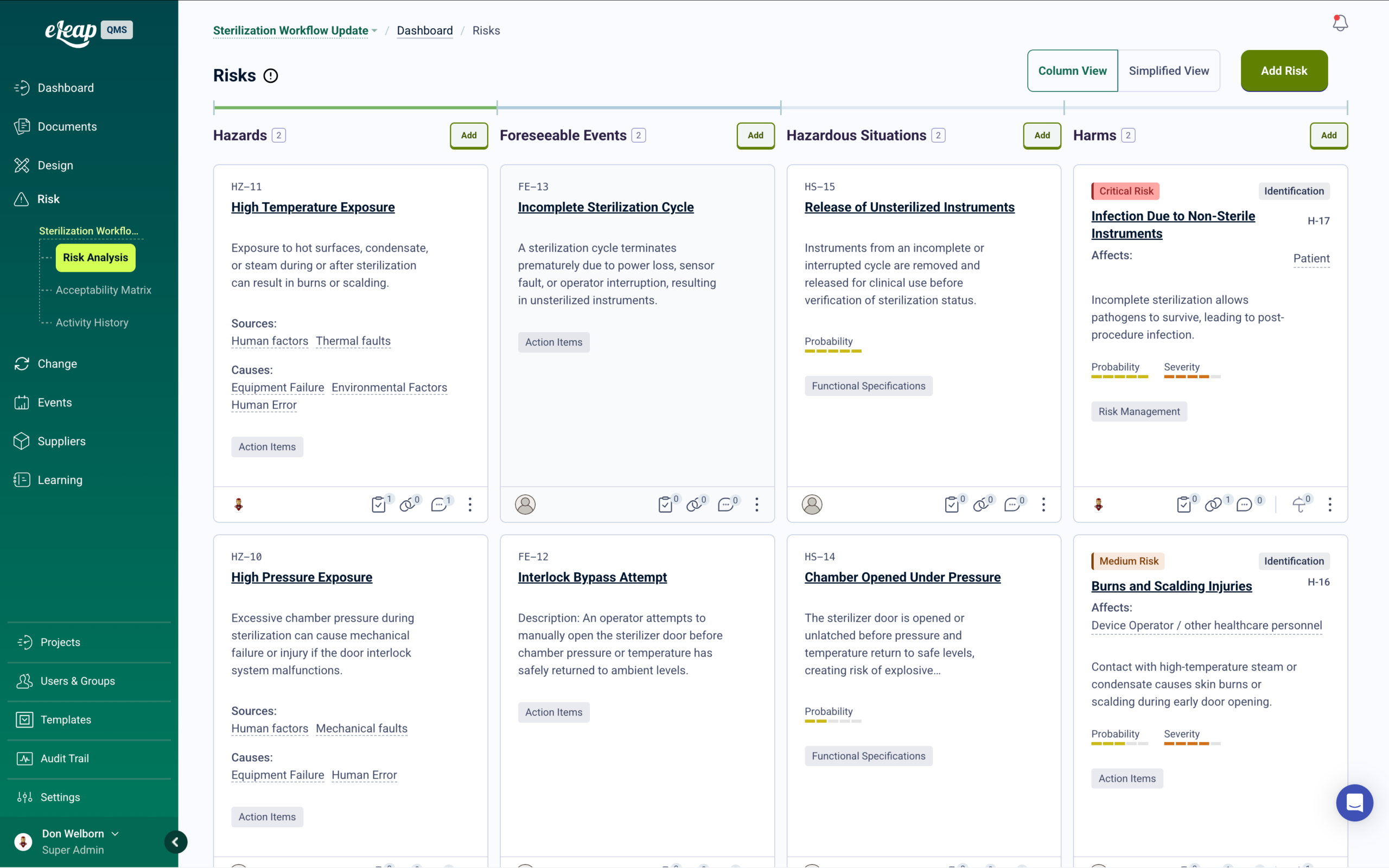
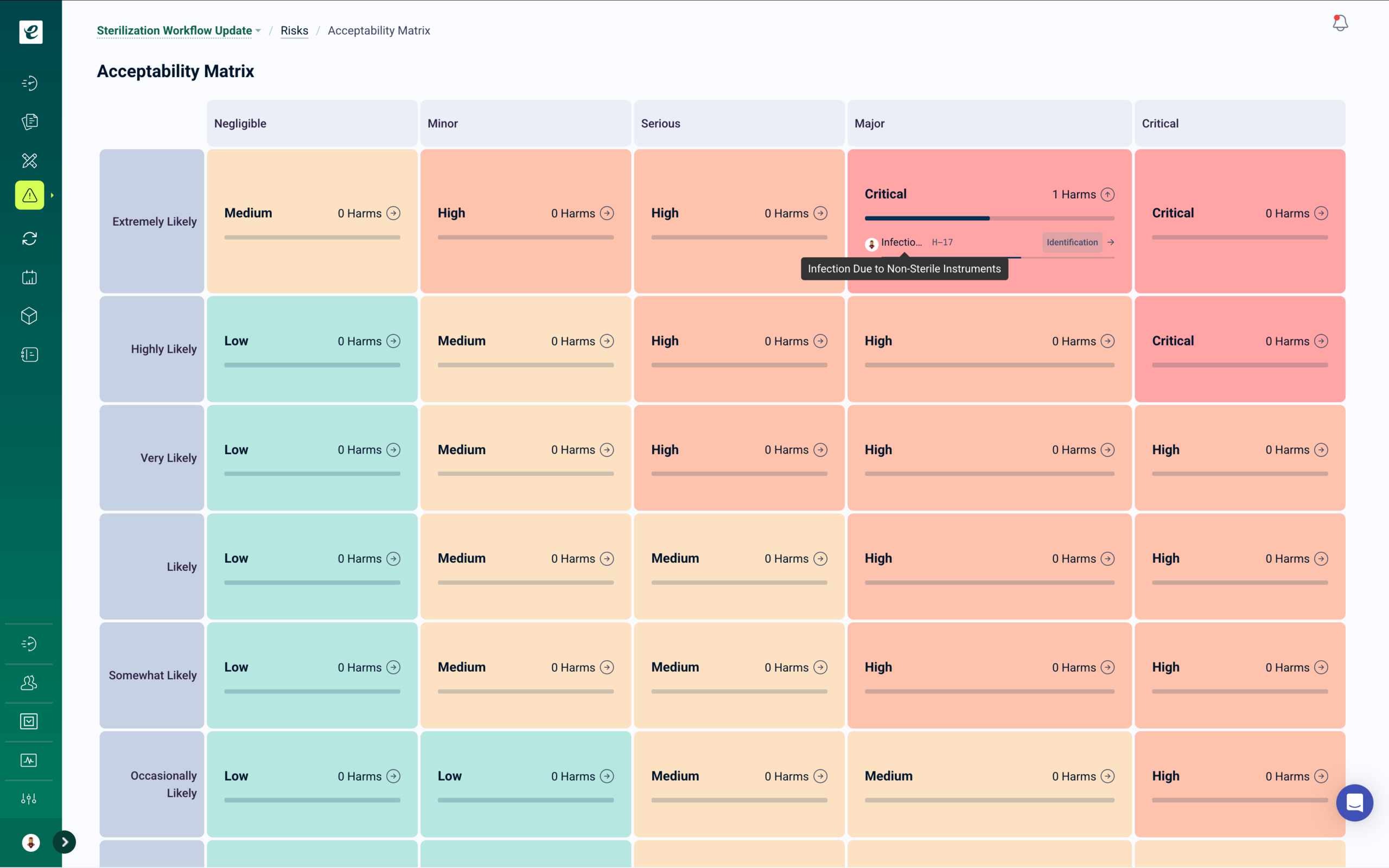
Integrated Risk Management
eLeaP’s integrated risk management module supports comprehensive assessments with customizable probability and severity scoring, risk priority numbers, and residual risk tracking. When risk assessments identify new hazards or control measures, targeted training is automatically deployed to affected personnel. Connect risk findings to CAPAs, design controls, and supplier management — with complete audit trails across every linked quality process.
Corrective and Preventive Actions (CAPA) transform quality management into a strategic endeavor. eLeaP’s integrated risk management system turns quality incidents into learning opportunities by identifying root causes and ensuring corrective actions prevent recurrence. Conduct ISO 14971-aligned risk assessments with scoring for probability and severity, and deploy targeted training for new control measures.
Root Cause Analysis Connected to Training and Performance Data
The root cause analysis feature connects quality events to training history, assessments, and performance records — helping identify whether issues stem from knowledge gaps, skill deficiencies, execution issues, or process failures. Monitor control effectiveness, track CAPA status from initiation to closure, and identify patterns across root causes using visual dashboards. Complete audit trails link every corrective action to the training and competency records that support it. Learn more.
Change Control with Automatic Training
Document changes, design updates, and process modifications automatically trigger targeted training for affected personnel — with escalation workflows ensuring nothing is missed.
eLeaP’s Change Control module manages the complete change lifecycle — from initiation through approval and implementation. When changes are approved, targeted training is automatically deployed to affected personnel based on their roles and responsibilities.
Manage change requests from initiation to implementation with tailored approval workflows.
Once changes are approved, training assignments are automatically deployed to affected personnel based on their roles and responsibilities. Smart assignment rules ensure the right people receive the right training, learning paths guide employees through required content, assessments verify competency, and completion records are maintained for audit review — so every change is fully documented from approval through verified retraining. Learn more.
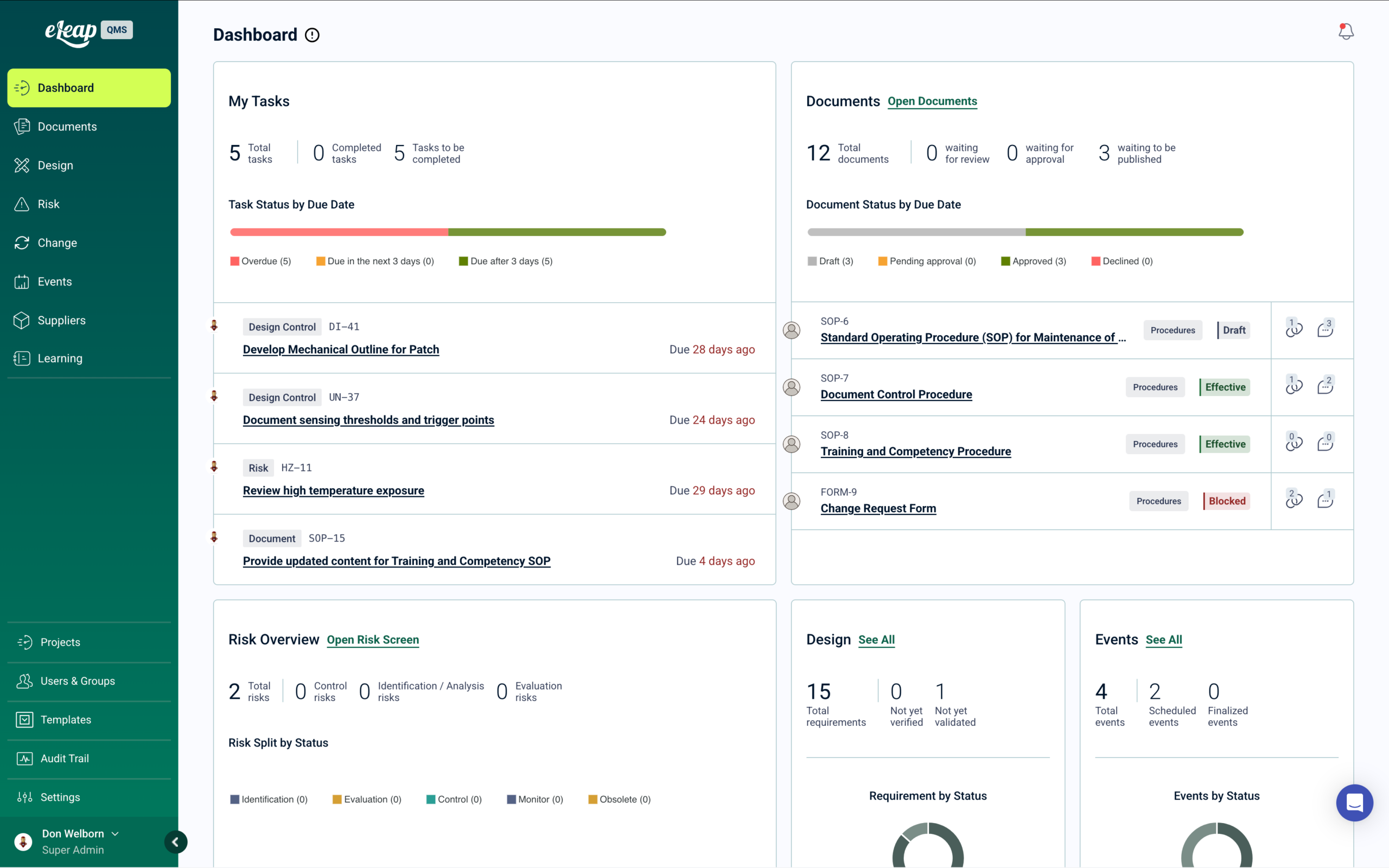
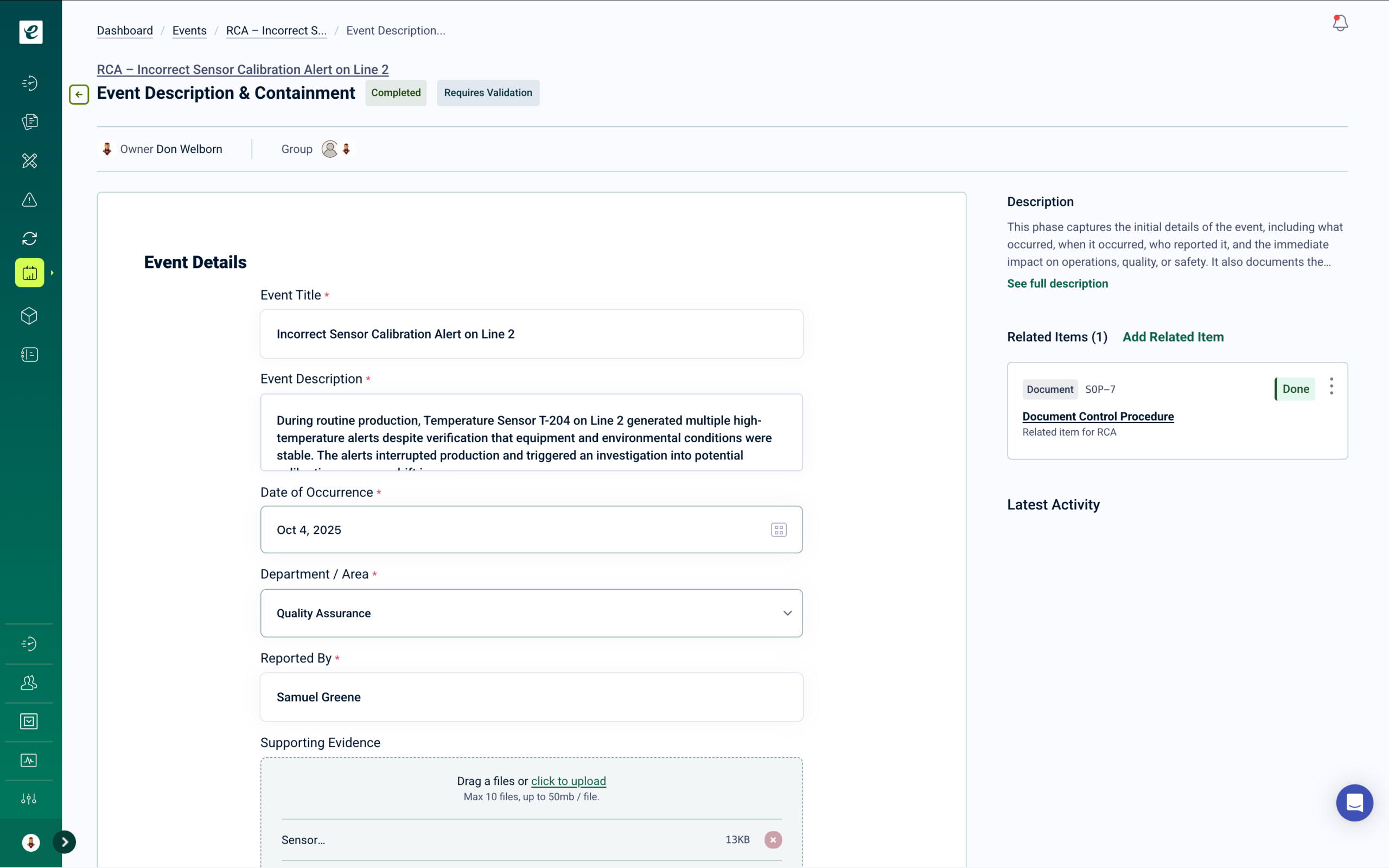
Quality Events Trigger Training Automatically
Every quality event — CAPAs, deviations, audit findings — automatically triggers targeted training assignments through the integrated Learning module.
Automated Training Assignment Across Every Quality Event
When quality events occur, the system automatically deploys training assignments to affected personnel based on their roles and responsibilities.
Smart assignment rules ensure the right people receive the right training, while learning paths and assessments guide employees through required content.
Training completion records are maintained automatically — so your team stays compliant and audit-ready without manual coordination. Learn more.
Supplier Quality Management
Risk-based supplier classification, audit management, and certificate tracking — with nonconformances and findings linked directly to your internal quality processes.
eLeaP’s Supplier Quality Management module manages the complete supplier lifecycle — tracking performance, audit results, compliance certifications, and quality agreements across your supply chain. Supplier findings and nonconformances are linked directly to your internal CAPA and change control processes, maintaining a connected quality system from supplier performance through organizational outcomes.
Supplier Performance Visibility Across Your Entire Supply Chain
Monitor supplier performance metrics, document audit results, and maintain compliance certifications with expiration tracking and automatic renewal notifications.
When supplier findings are identified, nonconformances are linked directly to your internal CAPA and change control processes — maintaining complete audit trails for regulatory review and connecting supply chain quality to your broader quality system. Learn more.

Unified Learning and Quality — Fully Integrated
A purpose-built platform for regulated industries where quality processes and training compliance operate as one connected system.
eLeaP integrates your QMS and LMS so quality events automatically trigger training assignments and competency verification is tracked in real time. Training completion links directly to quality outcomes — informing root cause analysis, closing corrective action loops, and maintaining continuous audit readiness across every module.
When quality events occur, the system automatically deploys targeted training and identifies whether issues stem from knowledge gaps, skill deficiencies, or process failures. Training completion updates compliance records, and competency assessments inform root cause analyses — creating a closed loop between quality events and verified corrective action.
This integrated approach supports faster issue resolution, more effective corrective actions, and measurable improvements in compliance outcomes — with complete audit trails connecting quality decisions to training records across every module.

Compliance Built Into Every Quality Workflow
eLeaP’s integrated QMS connects quality processes and training compliance across every module — so compliance is a natural outcome of daily quality activity rather than a separate administrative task. Quality teams and regulatory leadership get real-time visibility into competency status, training completion, and quality outcomes, supporting data-driven decisions and continuous improvement across the organization.
-
Automatic Training Triggers
Quality events, document changes, design updates, and risk assessments automatically deploy targeted training assignments to affected personnel — based on their roles and responsibilities.
-
Integrated Competency Verification
Assessments verify knowledge at every stage. Training completion links directly to quality outcomes, and competency records inform root cause analysis and corrective actions.
-
Audit-Ready Compliance
Complete audit trails connect quality decisions to training records across every module. Compliance documentation is maintained automatically, making regulatory readiness a natural outcome of integration.
Quality Excellence Through Integrated Learning
eLeaP’s integrated QMS gives quality teams and regulatory leadership real-time visibility into training completion, competency status, and quality outcomes across all seven modules. Quality leaders get the insights needed to make data-driven decisions, while team members have clear visibility into their training requirements and compliance status — supporting continuous improvement at every level of the organization. Learn more.
-
Continuous Training Integration
Quality incidents automatically deploy targeted training. Design changes cascade learning requirements. Risk assessments trigger competency verification. Every quality process is connected to a training outcome.
-
Connected Quality Data
All seven modules — Documents, Design & Development, Risk, Change, Events, Suppliers, and Learning — operate as one integrated system with complete audit trails connecting quality decisions to training records.
-
Competency-Driven. Root Cause Analysis
Root cause analysis connects training completion, competency assessments, and quality outcomes — so you can identify whether issues stem from knowledge gaps, skill deficiencies, or process failures with confidence.
-
Here, When You Need Us
Our quality experts are available to support your implementation, optimize your workflows, and ensure your team gets maximum value from the platform.