QMS for Biotechnology
The only eQMS that unifies cGMP quality processes and GxP training compliance in one integrated system

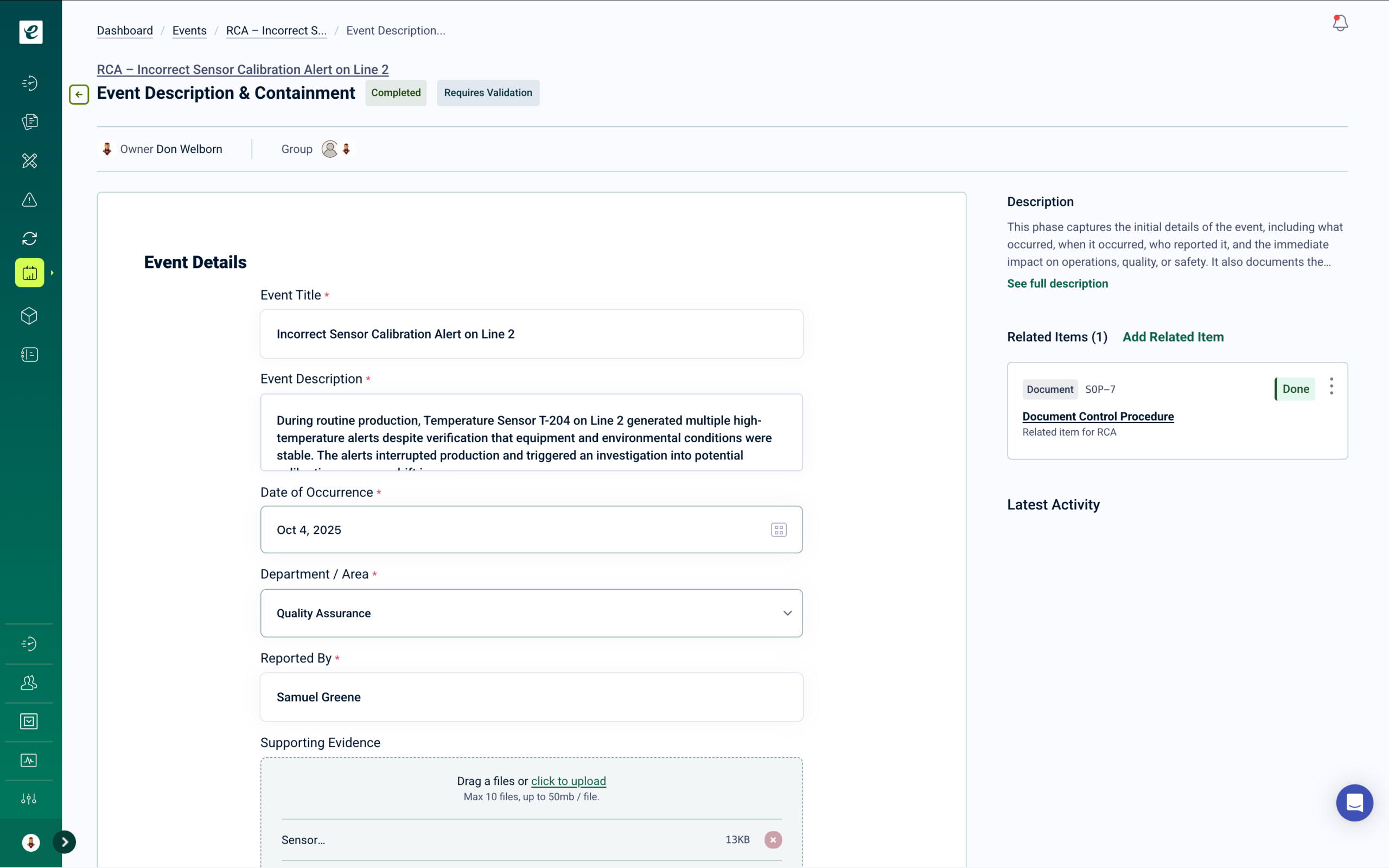
cGMP Quality Management for Biotechnology Operations
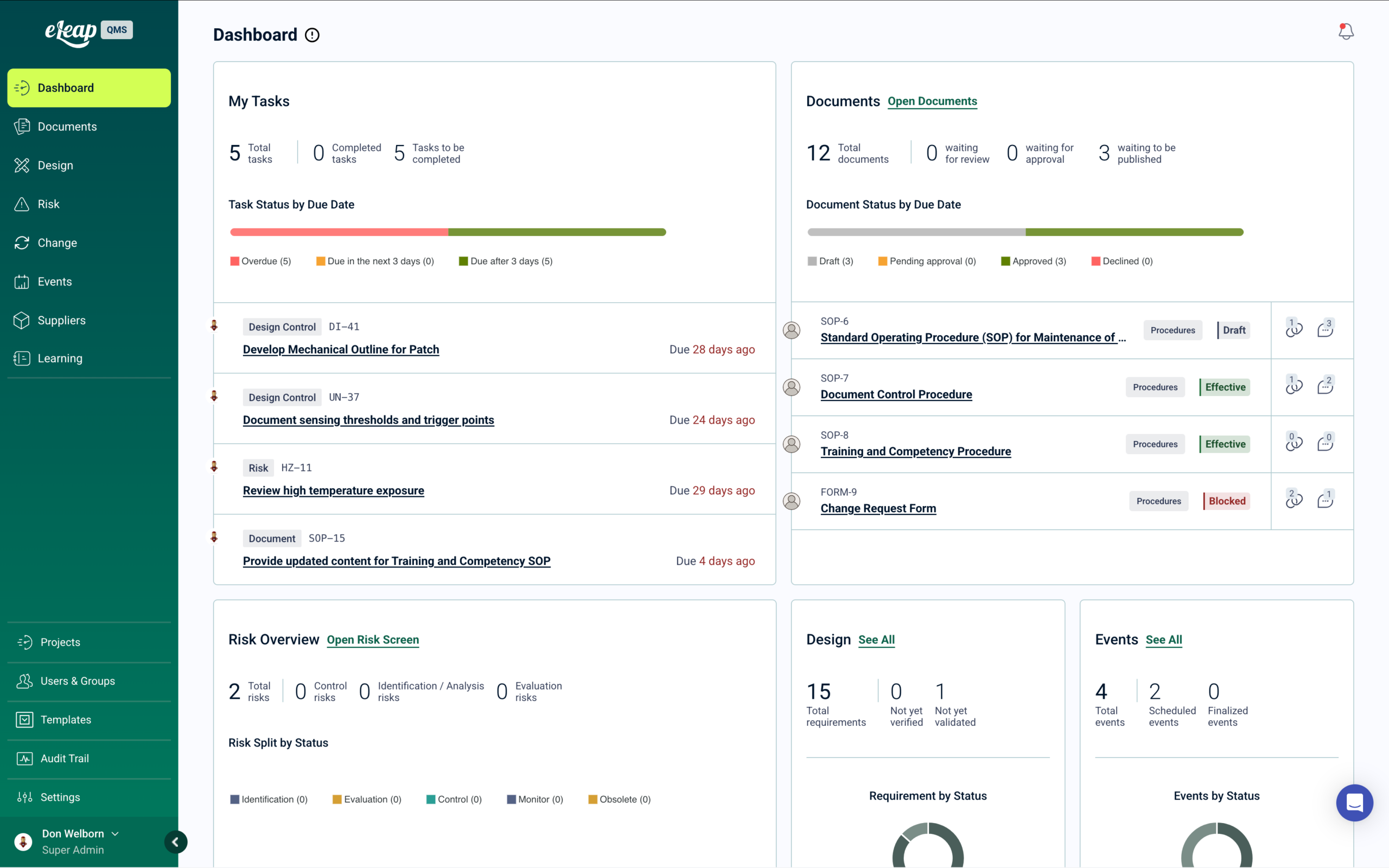
eLeaP is the only Quality Management System with enterprise Learning Management built in — purpose-built for biotechnology companies operating under cGMP, GxP, and ICH guidelines. From cell culture process documentation through BLA submissions, eLeaP automates personnel qualification across every quality process in a single pre-validated platform. → Get a Demo.
How It Works:
When SOPs are revised and approved, they automatically become training assignments with completion tracking. Process changes cascade required training to affected manufacturing roles. Risk assessments trigger mitigation training when control measures are updated. CAPAs and OOS investigations drive targeted corrective training with documented effectiveness verification — all traceable for FDA and international regulatory inspections.
Our platform deploys in weeks, with built-in 21 CFR Part 11 compliance and templates aligned to FDA, ICH, and GxP requirements. Trusted by biotech, biopharma, and life sciences organizations at every stage from startup to commercial manufacturing.
Controlled Document Management for cGMP Biotech Operations
SOPs, batch records, validation protocols, analytical methods, and regulatory submission documents — all managed in one Part 11-compliant system with automatic training deployment on approval.
Document Control That Scales with Your Development Stage
Biotech organizations manage a high volume of frequently revised documents across development, scale-up, and commercial manufacturing. eLeaP controls the full document lifecycle — from draft through approval through retirement — with version-controlled workflows, 21 CFR Part 11 electronic signatures, and complete audit trails that support both internal audits and regulatory submissions.
What makes eLeaP different: when a procedure goes effective, training automatically deploys to every affected role with no manual assignment required. Read-and-understood acknowledgments, comprehension assessments, and training matrices maintain themselves. When an FDA inspector or BLA reviewer requests training documentation, everything is in one place and traceable. Learn more. Get a Demo.

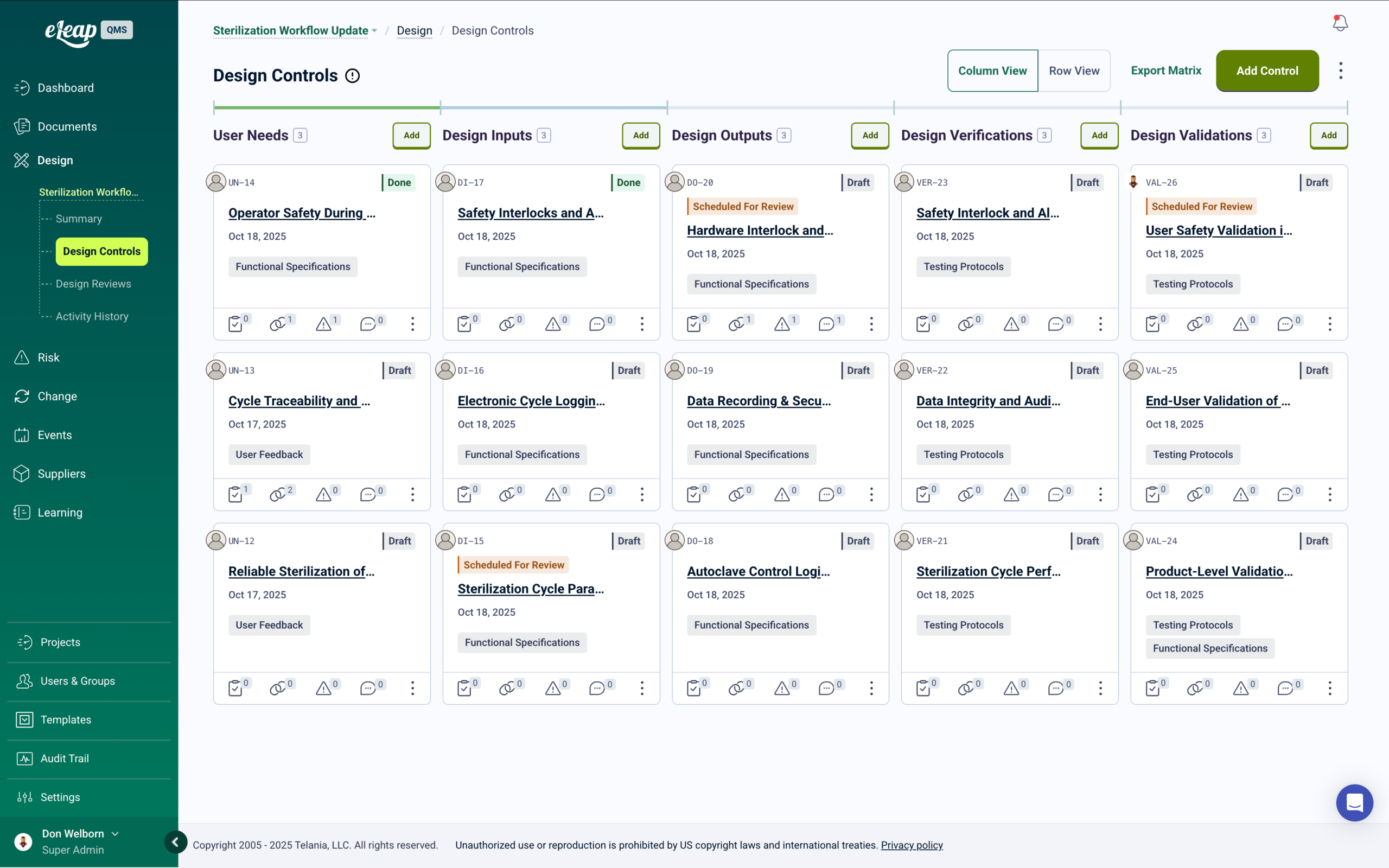
Process and Product Specification Management
Manage formulation specs, process parameters, raw material standards, and analytical method documentation through structured review and approval — with full version control and automatic training when specifications change.
Bioprocess and Product Specification Management
Cell culture parameters, fermentation conditions, purification specifications, and analytical method documentation — managed through structured review and approval with automatic training when specifications change.
Specification Control for Biological Manufacturing
Biologic manufacturing operates on the principle that the process is the product. Tight control of bioprocess specifications — for cell lines, media composition, process parameters, and product quality attributes — is central to regulatory compliance and product consistency. eLeaP manages the full specification lifecycle from creation through revision through retirement, with structured approval workflows and complete change history.
When specifications are revised, affected manufacturing and QA personnel receive automatic training assignments before the updated version goes effective. Version control prevents use of superseded specifications in production. Specification history, approval records, and associated training completions are maintained in a single traceable system.
Get a Demo.
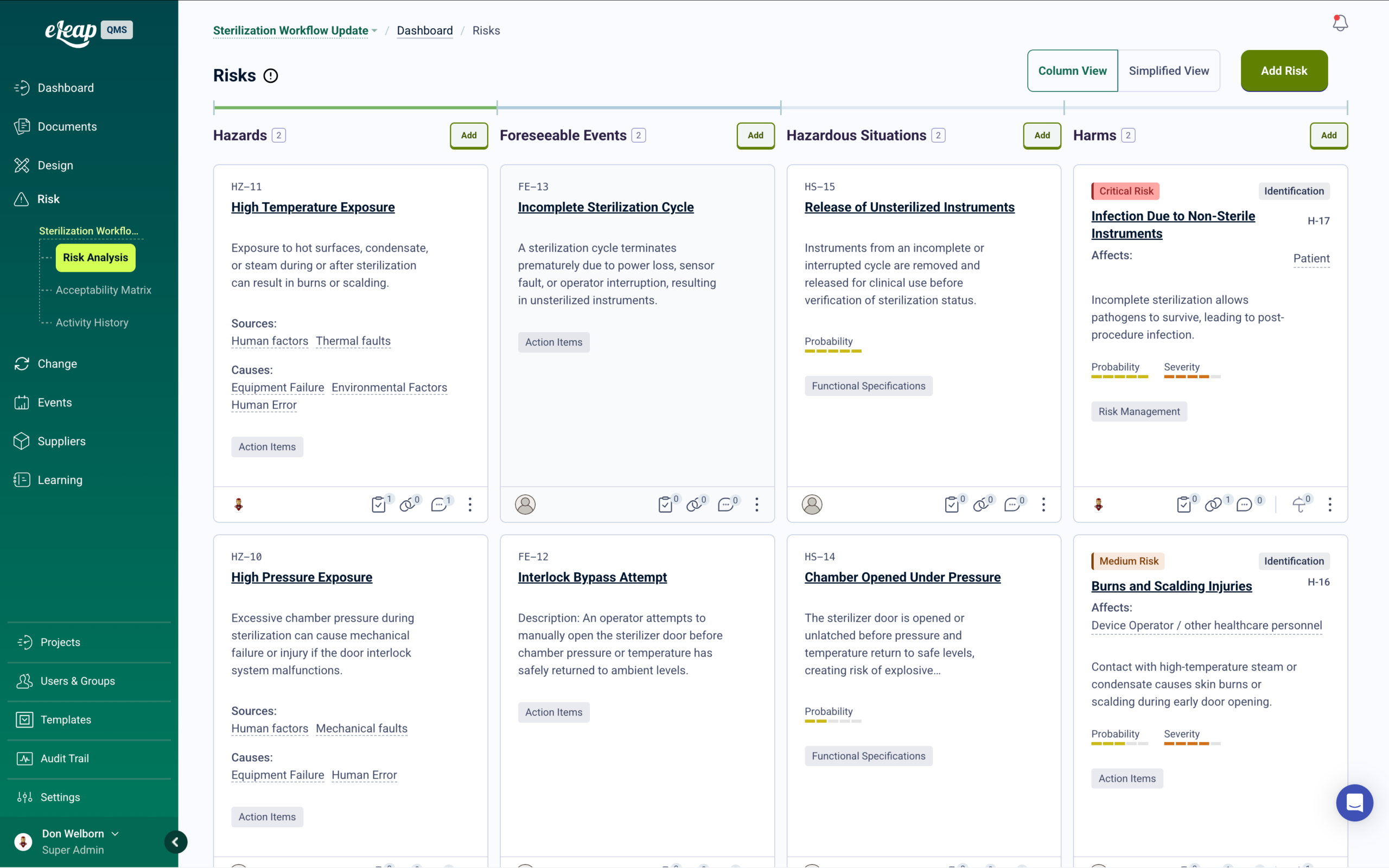
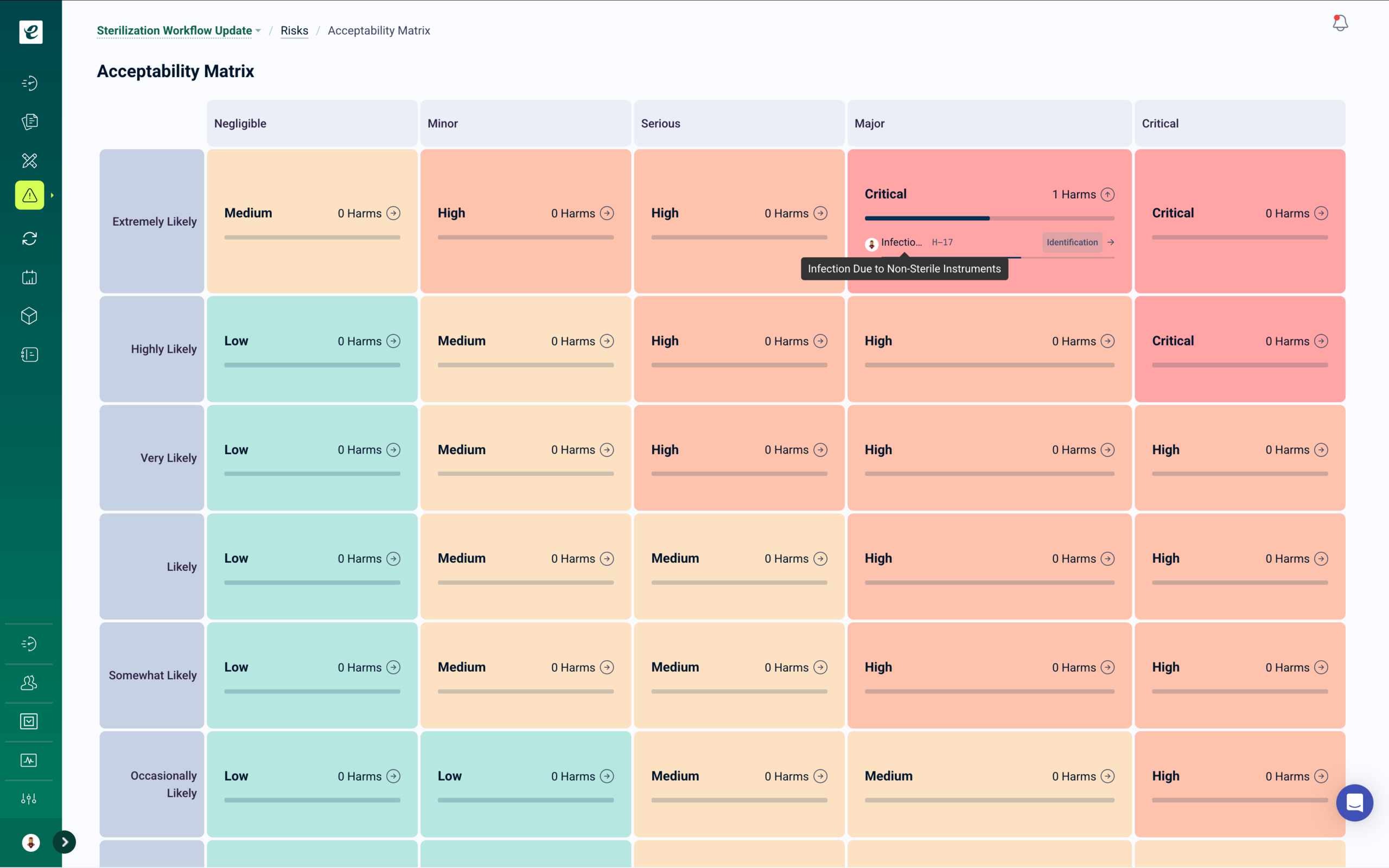
ICH Q9-Aligned Risk Management with Integrated Training
Risk assessments for biological processes, containment procedures, and product quality attributes — with automatic training deployment when control measures are introduced or updated.
eLeaP’s risk management module supports ICH Q9 risk methodology across biotechnology manufacturing operations. Conduct FMEAs for bioprocess steps, assess contamination and cross-contamination risks, and document control measures with probability and severity scoring. Path-to-Harm visualization provides complete risk traceability from hazard identification through control verification.
Risk-Based Quality Management for Biological Operations
When risk assessments identify new control measures or procedural safeguards, training automatically deploys to affected personnel before implementation. Root cause analysis connects quality events to training history, identifying whether issues stem from knowledge gaps, process variability, or equipment failures. Risk reviews, control effectiveness monitoring, and CAPA linkage are maintained in one traceable system. Get a Demo.
cGMP Change Control with Automatic Training
Cell line changes, process modifications, equipment qualifications, and procedure revisions automatically trigger training for affected personnel — with escalation workflows and verified implementation before changes go live.
In biotech manufacturing, change control must account for the sensitivity of biological processes — where process changes can affect product quality attributes in ways that require formal comparability assessment and updated regulatory submissions. eLeaP’s change control module manages the full lifecycle from request through implementation and verification, with automated training deployment built into the approval workflow.
Change Control Built for Biological Process Management
When a change is approved, training deploys immediately to all roles identified in the impact assessment. Competency assessments verify understanding before implementation. Change closure requires documented training completion, ensuring the gap between change approval and verified personnel qualification is eliminated. Complete change and training records are available for FDA inspection and regulatory submissions. Get a Demo.
CAPA, Deviation, and OOS Management
Quality events automatically identify training needs and deploy assignments — connecting investigations, corrective actions, and personnel competency in one traceable system.
From Quality Events to Verified Competency
Biotech operations generate deviations, out-of-specification results, contamination events, and yield failures that each require documented investigation and corrective action. eLeaP’s events management module handles CAPAs, deviations, OOS investigations, and audit findings — and automatically connects each to training when investigation identifies a personnel competency gap.
When a deviation identifies a procedural misunderstanding or an OOS result points to analyst error, training assignments deploy automatically. CAPA closure requires documented training completion and effectiveness verification. Audit findings link directly to corrective training actions with tracked closure. Every event, every investigation, every corrective action — connected, compliant, and audit-ready. Get a Demo.
Supplier Quality Management for Biotech Supply Chains
Qualification of raw material suppliers, contract testing labs, cell banking facilities, and CDMOs — with audit management and training triggers when supplier quality issues identify internal gaps.
Biotechnology supply chains include suppliers of critical raw materials (media, sera, growth factors), single-use components, reference standards, and contract services that directly affect product quality and regulatory compliance. eLeaP’s supplier management module supports supplier qualification workflows, quality agreement documentation, incoming material inspection records, and audit management.
cGMP Supply Chain Quality for Biological Manufacturing
When supplier audits identify gaps in internal receiving, testing, or qualification procedures, training automatically deploys to affected internal teams. Supplier performance metrics, qualification status, and audit findings are tracked in a system directly linked to your broader quality operations. Get a Demo.

GxP Training Compliance — Built In, Not Added On
Enterprise learning management is native to the platform. GxP training obligations are met through automatic triggers from quality events, change control, and document approvals — not managed in a separate system.
GxP training obligations in biotech span cGMP manufacturing, GLP laboratory operations, GCP clinical activities, and GVP pharmacovigilance functions. Meeting these obligations across a growing organization — with changing procedures, recurring quality events, and expanding product portfolios — requires more than a training system that talks to your QMS.
eLeaP’s learning management is native to the platform. SOPs approved in Document Management automatically create training assignments. CAPAs closed in Events automatically trigger effectiveness verification. Change Control approvals automatically deploy role-based training before implementation. Qualification matrices show who is trained on what, in real time, across every process, procedure, and piece of equipment. When a regulator or auditor asks for training records, you answer immediately — from one system.

Building GxP Compliance Into Every Workflow
Regulatory inspections of biotech facilities consistently cite training deficiencies, inadequate investigation depth, and gaps between quality events and corrective training. eLeaP eliminates these gaps by making training an automatic output of every quality process — not a manual step requiring coordination between systems.
-
Automatic Training Triggers
Procedure approvals, change implementations, deviation closures, and CAPA actions automatically deploy training to the right personnel at the right time.
-
Real-Time Qualification Verification
Personnel qualification status is visible across all processes, procedures, and equipment — no spreadsheets, no manual tracking.
-
Inspection-Ready by Design
Complete audit trails connect quality decisions to training records. When inspectors ask, you answer immediately.
Quality and Training Working as One System
Biotech quality management should accelerate your development timelines — not slow them down. eLeaP gives quality leaders the tools to eliminate compliance blind spots while giving manufacturing and laboratory personnel timely, relevant training connected directly to their work. Get a Demo.
-
Continuous Training Integration
Every quality process — documents, specifications, risk, change, events, suppliers — automatically drives training when it matters.
-
Connected Quality Data
All seven modules work as one system. No disconnected records. No manual handoffs. Complete audit trails.
-
Competency-Driven. Root Cause Analysis
Investigations connect training history, competency assessments, and quality outcomes — identifying whether issues stem from knowledge gaps or process failures.
-
Here, When You Need Us
Our quality experts support implementation, process optimization, and compliance program development for biotech organizations at every stage.